N-Acetylglucosaminyltransferase III (GnT-III) catalyzes the transfer of GlcNAc from UDP-GlcNAc to the core β-mannose of N-glycans via β1,4-linkage (Fig 1). The transferred GlcNAc residue is referred to as a bisecting GlcNAc. The addition of the bisecting GlcNAc residue to the core β-mannose by the enzyme prevents the action of other GlcNAc transferases involved in the biosynthesis of multiantennary sugar chains, leading to a decrease in the branch formation of N-glycans. Because of this regulatory role, the enzyme has been considered to be a key glycosyltransferase in N-glycan biosynthesis (Ikeda Y, 2002).
GnT-III activity can be assayed using a sensitive and convenient method involving a fluorescent-labeled oligosaccharide acceptor, pyridylaminated agalacto-biantennary sugar chain (PA-GnGnbi). In this assay system, a product, PA-bisected GnGnbi, can be separated from the unreacted acceptor substrate and quantitated using reversed phase HPLC with a fluorescence detector. |
| Category | Glycosyltransferases & related proteins |
| Protocol Name | Enzyme assay of N-acetylglucosaminyltransferase III (GnT-III) |
Authors
 |
Korekane, Hiroaki
Department of Disease Glycomics (Seikagaku Corporation), The Institute of Scientific and Industrial Research, Osaka University
Taniguchi, Naoyuki
*
Systems Glycobiology Research Group, Chemical Biology Department, RIKEN Advanced Science Institute
*To whom correspondence should be addressed.
|
Reagents
 |
| ● |
100 μM PA-GnGnbi in Milli-Q water |
| ● |
2× GnT-III reaction buffer: 250 mM MES-NaOH buffer, 40 mM UDP-GlcNAc, 20 mM MnCl2, 400 mM N-acetylglucosamine, 1% (w/v) Triton X-100, pH 6.25. Stored at −20°C until use. |
|
Instruments
 |
| ● |
TSKgel ODS-80TM column (0.46 × 15 cm, Tosoh Corp., Tokyo, Japan) |
|
| Methods |
|
1. |
Preparation of crude enzyme extracts from tissues |
| 1) |
Homogenize tissues with 4 vol of 10 mM Tris-HCl (pH 7.4), 0.25 M sucrose, and proteinase inhibitors (Rosch, Complete proteinase inhibitors cocktail EDTA-free). |
Comment 0
|

|
| 2) |
Centrifuge at 600 × g for 5 min at 4°C. |
Comment 0
|

|
| 3) |
Collect Sup and then use for the enzyme activity assay. |
Comment 0
|
|
|
|
2. |
Preparation of crude enzyme extracts from cultured cells |
| 1) |
Wash cultured cells with PBS (−) 3 times and then harvest them as cell pellets. |
Comment 0
|

|
| 2) |
Suspend the cell pellets in 100–200 μL of ice-chilled PBS (−). |
Comment 0
|

|
| 3) |
Sonicate the suspended cells for 5–10 min with a Bioruptor, and then use for the enzyme activity assay. |
Comment 0
|
|
|
|
3. |
|
| 1) |
Mix the following components in a small plastic tube.
2× GnT-III reaction buffer 5 μL
100 μM GnGnbi-PA 1 μL
Enzyme extracts 4 μL
Final 10 μL |
Comment 0
|

|
| 3) |
Add 40 μL of Milli-Q water and then boil for 2 min to stop the enzyme reaction. |
Comment 0
|

|
| 4) |
Centrifuge at 20,000 × g for 5 min to remove denatured proteins and then collect Sup. |
Comment 0
|

|
| 5) |
Analyze 10 μL of the cleared Sup using reversed phase HPLC (Fig. 2) and then calculate the enzyme activity from the product peak area. |
Comment 0
|
|
|
| Notes | For reagents :
the PA-acceptor oligosaccharide is obtained from hen egg yolk by means of purification of sialylglycopeptide (SGP) (Seko A, 1997), hydrazinolysis, re-N-acetylation, and pyridylamination (Natsuka S, 1998). Through sequential exoglycosidase digestions of the PA-SGP with sialidase and β-galactosidase, the acceptor oligosaccharide PA-GnGnbi is obtained. The PA-GnGnbi is further purified by reversed phase HPLC using a TSKgel ODS-80TM column (0.78 × 30 cm, Tosoh Corp.), which has been equilibrated with 20 mM ammonium acetate buffer pH 4.0, containing 0.25% (v/v) 1-butanol, at a flow rate of 2 mL/min, at 55°C. Elution is performed isocratically and the fluorescence of the column eluate is detected at excitation and emission wavelengths of 320 and 400 nm, respectively. |
| Figure & Legends |
Figure & Legends 
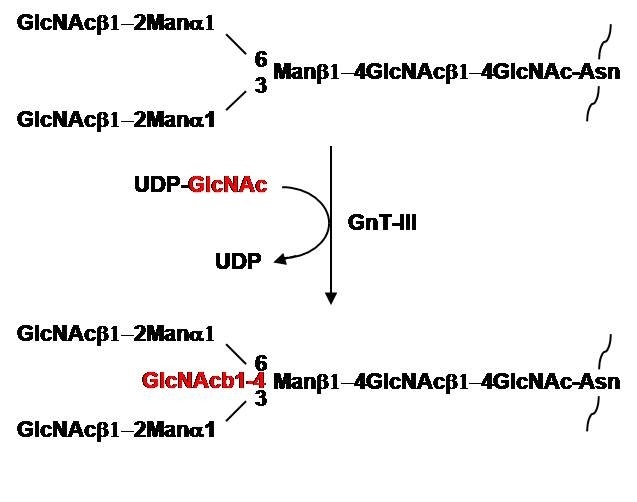
Fig. 1. Reaction catalyzed by GnT-III

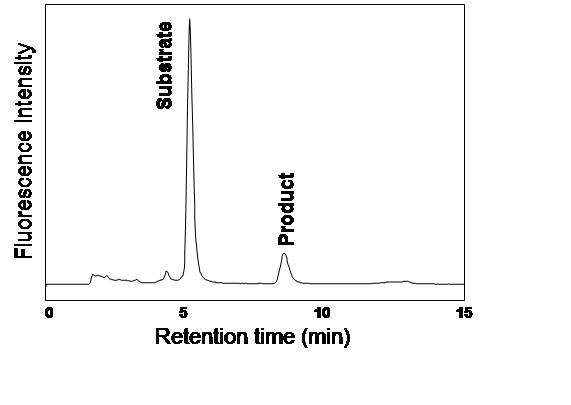
Fig. 2. A typical chromatogram for the enzyme assay
Column; TSKgel ODS-80TM (0.46 × 15 cm)
Buffer A; 20 mM ammonium acetate pH 4.0
Buffer B; buffer A containing 1% (v/v) 1-butanol
Elution; 25% buffer B, isocratic
Flow rate; 1 mL/min
Column temp; 55℃
Fluorescence, excitation/emission=320/400 nm |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2015-08-05 14:53:46 |
- Ikeda, Y., and Taniguchi, N. (2002) N-Acetylglucosaminyltransferase-III. In: Taniguchi N, Honke K, Fukuda M, editors. Handbook of Glycosyltransferases and Related Genes. Tokyo: Springer. p. 80–86.
- Seko, A., Koketsu, M., Nishizono, M., Enoki, Y., Ibrahim, H.R., Juneja, L.R., Kim, M., and Yamamoto, T.(1997) Occurrence of a sialylglycopeptide and free sialylglycans in hen’s egg yolk. Biochim. Biophys. Acta. 1335, 23–32 [PMID : 9133639]
- Natsuka S, Hase S. 1998. Analysis of N- and O-glycans by pyridylamination. Methods Mol Biol. 76, 101–113 [PMID : 9664349]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Korekane, Hiroaki,
Taniguchi, Naoyuki,
(2015). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.21,9,2024 .
How to Cite this Work in Website:
Korekane, Hiroaki,
Taniguchi, Naoyuki,
(2015).
Enzyme assay of N-acetylglucosaminyltransferase III (GnT-III).
Retrieved 21,9,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t43.
html source
Korekane, Hiroaki,
Taniguchi, Naoyuki,
(2015).
<b>Enzyme assay of <em>N</em>-acetylglucosaminyltransferase III (GnT-III)</b>.
Retrieved 9 21,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t43" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t43</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|