Gas-liquid chromatography (GC) is a conventional method to quantify the carbohydrates in samples. This chemical method is one of the most reliable methods if enough amounts of sample are available. The monosaccharide analysis by GC consists of three steps: (i) Quantitative solvolysis of carbohydrates into monosaccharide components; (ii) Volatilization of the monosaccharides by trimethylsilylation; and (iii) Separation and quantitation of the monosaccharides as the trimethylsilylated derivatives on GC. |
| Category | Isolation & structural analysis of glycans |
| Protocol Name | Carbohydrate analysis by gas-liquid chromatography |
Authors
 |
Sato, Chihiro
*
Bioscience and Biotechnology Center, Nagoya University
Kitajima, Ken
Bioscience and Biotechnology Center, Nagoya University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Myo-inositol as an internal standard (Wako Pure Chemical Industries, Ltd. Osaka, Japan) |
| ● |
0.5N HCl-MeOH (Kokusan Kagaku Co., Ltd. Tokyo, Japan) |
| ● |
Absolute MeOH (Sigma-Aldrich, St. Louis, MO) |
| ● |
Acetic anhydride (Sigma-Aldrich) |
| ● |
Absolute pyridine (Sigma-Aldrich) |
| ● |
Hexamethyldisilazane(HMDS) (Wako Pure Chemical Industries, Ltd.) |
| ● |
Trimethylsilyl chloride (TMSC) (Wako Pure Chemical Industries, Ltd.) |
|
Instruments
 |
| ● |
GLC (GC-14A: Shimadzu Corp., Kyoto, Japan) |
|
| Methods |
|
1. |
|
| 1) |
Sample (1–10 µg) with an internal standard (1–10 µg). |
Comment 0
|

|
| 2) |
Dry up with rotary evaporator and then in a desiccator in vacuo (over P2O5). |
Comment 0
|

|
| 3) |
Add 0.5 mL of 0.5 N HCl-methanol, and heated at 65°C for 16 h. |
Comment 1
|

|
| 4) |
Dry up with evaporator and in the desiccator in vacuo (over P2O5). |
Comment 0
|

|
|
|
|
2. |
|
| 1) |
Add 0.5 mL of absolute MeOH, 10 µL of absolute pyridine, and 50 µL of acetic anhydride to the sample, and leave at room temperature for 15min. |
Comment 0
|

|
| 2) |
Dry up with N2-gas and then in a desiccator (over NaOH). |
Comment 0
|

|
| 3) |
Methylglycosides with or without re-N-acetylation |
Comment 0
|
|
|
|
3. |
|
| 1) |
Add 50 μL of absolute pyridine, 10 μL of HMDS and 5 μL of TMSC to the sample and leave at room temperature for 30 min. |
Comment 0
|

|
| 2) |
Centrifuge at 3,000 rpm for 10 min. |
Comment 1
|

|
| 3) |
Transfer the supernatant into a small glass tube. |
Comment 0
|

|
| 5) |
Solubilize with 50 μL of hexane. |
Comment 0
|

|
|
|
|
4. |
|
| 1) |
Inject 2–10 μL of the hexane solution into GLC.
Condition: for example: 140°C (10 min)・140°C–260°C (5°C/min)・260°C (10 min)
Detection FID, Column, CBJ-5 (30 m × 0.32 mm) |
Comment 0
|
|
|
| Notes | 1. If samples contain a large amount of lipids, you had better to perform hexane extraction procedure as described below before re-N-acetylation step.
1) methanolyzed sample
↓ add 0.5 mL of hexane and mix.
↓ centrifuge at 3000 rpm for 5 min.
2) methanol layer
↓ dry up with rotary evaporator and then in the desiccator (over P2O5) |
| Initial amount | |
| Discussion | Carbohydrates have to be derivatized for volatilization before application to GC, because they are involatile. Trimethylsilylation is often used for volatilization due to the facileness. Trimethylsilylated monosacharides can be separated on GC, and detected by flame ionization detector or mass spectrometry (MS) like electron impact MS. Methylation is also used for volatilization of carbohydrates; however, it is more often applied to the linkage analysis of glycosides. |
| Figure & Legends |
Figure & Legends

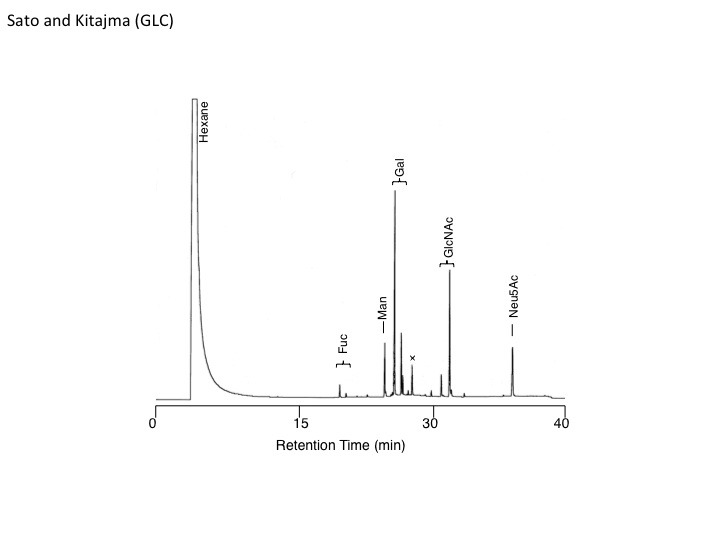
Fig. 1. Gas liquid chromatogram of Medaka hyosophorin.
Carbohydrate composition of N-linked glycan chain derived from hyosophorin sample was analyzed by GLC (Column: DB-5, Initial Temp: 140°C, Initial Time: 10 min, Final time: 260°C, Programmed rate 5°C/min). |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2015-03-24 13:52:16 |
- Nomoto, H., Iwasaki, M., Endo, T., Inoue, S., Inoue, Y., and Matsumura, G. (1982) Structures of carbohydrate units isolated from trout egg polysialoglycoproteins: short-cored units with oligosialosyl groups. Arch. Biochem. Biophys. 218, 335–341 [PMID : 7149739]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Sato, Chihiro,
Kitajima, Ken,
(2015). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.7,4,2025 .
How to Cite this Work in Website:
Sato, Chihiro,
Kitajima, Ken,
(2015).
Carbohydrate analysis by gas-liquid chromatography.
Retrieved 7,4,2025 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t223.
html source
Sato, Chihiro,
Kitajima, Ken,
(2015).
<b>Carbohydrate analysis by gas-liquid chromatography</b>.
Retrieved 4 7,2025 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t223" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t223</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|