Sugar-binding ability of lectins, especially those from mammals, is weak and animal lectins usually have Kd value approximately 10-4 M. Animal lectins including lectin-like receptors are far less abundantly expressed in cells when compared with plant lectins. Thus, it is quite difficult to evaluate sugar-binding activity and specificities in nature. To overcome such a characteristic of sugar-protein interaction, we established tetramerization of lectins by introducing biotin-tag sequence in combination with biotin ligase BirA1). Alternatively, lectin cell surface display method, in which lectin domain is expressed as a transmembrane fusion protein, is very useful especially for lectins that could not be prepared as soluble recombinant proteins expressed in E. coli. When CD3ζ is used as a cytoplasmic tail of a fusion protein, binding of sugar ligands through lectin domain causes phosphorylation of CD3ζ in cytoplasm, which resulted in transcription of a reporter gene (β-galactosidase, green fluorescence protein etc.) under the control of interleukin-2 responsive element (Fig. 1). By using the cell having such reporter gene, sugar-binding activity of lectins expressed on the cell surface sensitively measured without their purification. The reporter cell expressing a lectin domain on the cell surface also could be both an effective immunogen and a screening probe for establishment of specific monoclonal antibodies 2). In this chapter, preparation of a reporter cell expressing a lectin domain on cell surface and reporter assay for measuring sugar-binding ability of cell surface-expressing lectins. |
| Category | Sugar binding proteins |
| Protocol Name | Analysis of intracellular lectins using reporter cell expressing lectin on its surface |
Authors
 |
Yamamoto, Kazuo
Department of Integrated Biosciences, Graduate School of Frontier Sciences, The University of Tokyo
|
| KeyWords |
|
Reagents
 |
| ● |
reporter cells, BWZ.36 or 2B4. |
| ● |
transfection reagent: ripofectamine 2000, transfectam etc. |
| ● |
polyacrylamide-based sugar polymer (Glycotech, Gaithersburg, MD) or glycoproteins. |
| ● |
mono, di, oligosaccharides, and glycopeptides |
| ● |
|
| ● |
chlorophenol red-β-D-galactopyranoside (Wako Pure Chemical Industries Ltd., Osaka, Japan) |
|
Instruments
 |
| ● |
ELISA plate reader (Hitachi, Ltd., Tokyo, Japan) |
|
| Methods |
|
1. |
Preparation of reporter cells expressing lectin fusion protein on their surface |
| 1) |
Construct an expression plasmid containing cDNAs encoding the CD8β signal sequence followed by a Myc-tag, a lectin domain of desired protein, the NKp46 stalk domain, the CD8α transmembrane domain, and mouse CD3ζ. |
Comment 1
|

|
| 2) |
Transform mouse reporter cells, BWZ.36 or 2B4, with above plasmid using transfection reagent. |
Comment 1
|

|
| 3) |
Forty-eight hours after the transfection, harvest transfected cells and apply for the following reporter assay. |
Comment 0
|
|
|
|
2. |
Reporter assay for sugar binding |
| 1) |
Coat each well of ELISA plate with 50 μL of 20 μg/mL polyacrylamide-based sugar polymers, glycoproteins, anti-Myc antibody and so on. |
Comment 1
|

|
| 2) |
Culture reporter cells expressing lectin fusion protein (105 cells/well) on a sugar polymer-coated well at 37°C for 16 h in culture medium. |
Comment 0
|

|
| 3) |
Remove culture medium and then add 2.5 mM chlorophenol red-β-D-galactopyranoside as the substrate. |
Comment 0
|

|
| 4) |
Incubate for a few minutes at 37°C or a few hours at room temperature. |
Comment 1
|

|
| 5) |
Measure the colorimetric absorbance at 574 nm using ELISA plate reader. |
Comment 0
|
|
|
|
3. |
Inhibition assay using reporter cell |
| 1) |
Prepare several concentrations of inhibitory sugar solution.
Add inhibitory sugars in sugar polymer-coated wells and culture reporter cells at 37°C for 16 h. |
Comment 0
|

|
| 2) |
Remove culture medium and then add 2.5 mM of chlorophenol red-β-D-galactopyranoside as the substrate. |
Comment 0
|

|
| 3) |
Incubate for a few minutes at 37°C or a few hours at room temperature. |
Comment 1
|
|
|
| Initial amount | One microgram of sugar polymer/well
105 transfected cells/well (when transfection efficiency is approximately 30%) |
| Discussion | To discuss about sugar-binding specificity of lectins or to rule out nonspecific binding, reporter assay should be performed under several control conditions. When sugar-binding ability of expressed lectin fusion protein is quite strong, β-galactosidase expression level will reach a plateau. In this case, sugar polymer concentration should be considered to minimize cross-linking signaling to reporter cells. In the case of GFP-expressing reporter cell, 2B4, expression of GFP is measured by flow cytometry using FACScalibur and accompanying CellQuest software (BD Biosciences, San Jose, CA) and values of mean fluorescent intensity are compared. |
| Figure & Legends |
Figure & Legends

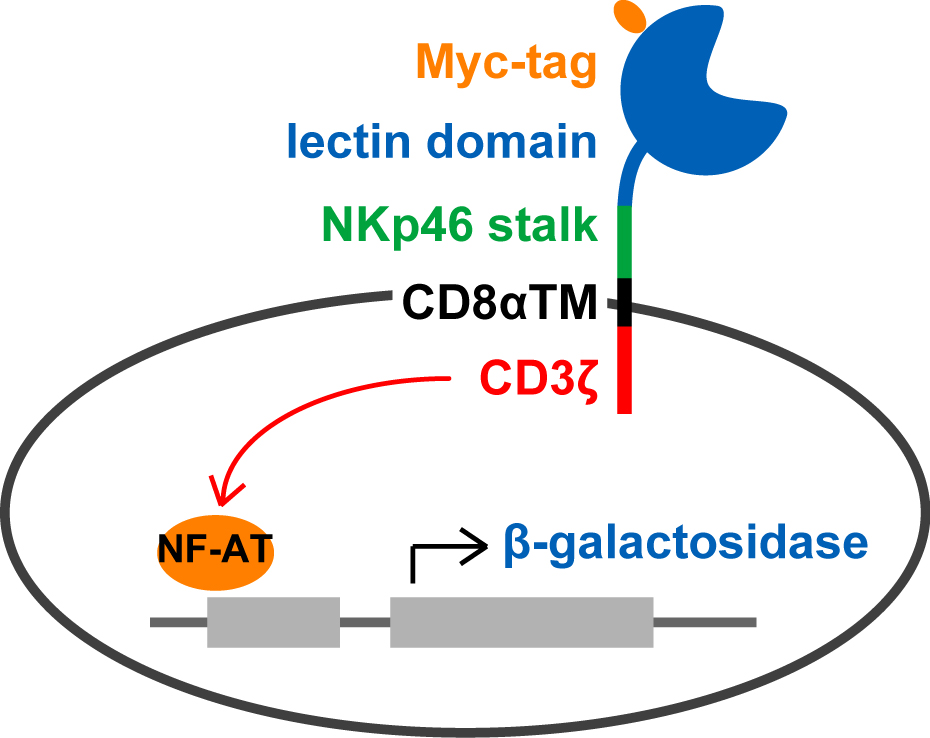
Fig. 1. Schematic illustration of reporter cells expressing lectin fusion protein on their surface. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-07-30 10:32:33 |
- Yamamoto, K., and Kawasaki, N. (2010) Detection of weak-binding sugar activity using membrane-based carbohydrates. Methods Enzymol. 478, 233–240 [PMID : 20816483]
- Qin, S., Kawasaki, N., Hu, D., Tozawa, H., Matsumoto, N., Yamamoto, K. (2012) Subcellular Localization of ERGIC-53 under endoplasmic reticulum-stress condition. Glycobiology 22, 1709–1720 [PMID : 22821029]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Yamamoto, Kazuo,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.4,5,2024 .
How to Cite this Work in Website:
Yamamoto, Kazuo,
(2014).
Analysis of intracellular lectins using reporter cell expressing lectin on its surface.
Retrieved 4,5,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t203.
html source
Yamamoto, Kazuo,
(2014).
<b>Analysis of intracellular lectins using reporter cell expressing lectin on its surface</b>.
Retrieved 5 4,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t203" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t203</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|