Permethylation is the most general derivatization used for the MS analysis of glycans. Hydrophilic glycans can be converted to hydrophobic molecules by permethylation. This conversion is useful for ionizing glycans in MS analysis, as well as for the separation of glycans from impurities such as salts. Hakomori’s method, which employs methylsulfenyl carbanion, was used for this type of derivatization 1) in previous times, although this has been supplanted by Ciucanu’s method, which employs NaOH and MeI in DMSO, for glycomics analysis 2) 3). Here, an example of a permethylation protocol according to the Ciucanu’s method is introduced. |
| Category | Isolation & structural analysis of glycans |
| Protocol Name | Permethylation for glycan analysis |
Authors
 |
Kameyama, Akihiko
Biotechnology Research Institute for Drug Discovery, Department of Life Science and Biotechnology, National Institute of Advanced Industrial Science and Technology (AIST)
|
| KeyWords |
|
Reagents
 |
| ● |
Iodomethane, Wako Special Grade, (Wako Pure Chemical Industries Ltd., Osaka, Japan) |
| ● |
Dimethyl sulfoxide, Infinity Pure Grade, (Wako Pure Chemical Industries Ltd.) |
| ● |
Sodium hydroxide, purum p.a., ≥98.0% (T), beads (small), (Fluka, Deisenhofen, Germany) |
|
Instruments
 |
| ● |
|
| ● |
Small glass tube with a screw cap, conical bottom |
| ● |
|
| ● |
Solid phase extraction device (Sep-Pak C-18; Waters Corp., Milford, MA) |
|
| Methods |
|
1. |
Permethylation for glycan analysis
* This procedure describes the small scale permethylation of glycans (<50 μg). |
| 1) |
An aliquot of glycan solution was transferred to a small glass tube*, and then evaporated to dryness using a centrifugal evaporator. |
Comment 1
|

|
| 2) |
A small amounts of sodium hydroxide and DMSO containing 1% (vol/vol) distilled water* were placed in an agate mortar, and grinded to a slurry. (Fig. 1) |
Comment 1
|

|
| 3) |
Ca. 50 μL of the slurry was transferred to the glass tube containing the glycans*. (Fig. 2 a and b) |
Comment 1
|

|
| 4) |
Ca. 50 μL of methyl iodide* was added to the glass tube. (Fig. 2 c) |
Comment 1
|

|
| 5) |
The tube was securely capped and the mixture vigorously shaken at high speed using a vortex mixer at room temperature for 15 min*. |
Comment 1
|

|
| 6) |
1% HCl (1 mL) was slowly added to the reaction mixture*, and then the mixture was applied to an Sep-Pak C-18 (50 mg, syringe type:1 cc) cartridge**. |
Comment 1
|

|
| 7) |
The Sep-Pak Cartridge was washed with distilled water (3 mL). |
Comment 0
|

|
| 8) |
After the washing, the permethylated glycans were eluted with 80% (vol/vol) acetonitrile (1 mL), and evaporated to dryness by centrifugal evaporator. |
Comment 0
|
|
|
| Notes | Safety concerns
This procedure should be performed in a fume hood.
Wear suitable protective eyewear and gloves. Do not use latex gloves, which are permeable to methyl iodide. Nitrile or poly chloroprene gloves are recommended.
Acknowledgement
Author thanks Drs K-H. Khoo and S-Y. Yu for training on this practical handling for permethylation. |
| Figure & Legends |
Figure & Legends 

Fig. 1. Preparation of NaOH slurry
(a) small beads of NaOH in an agate mortar, (b) addition of DMSO containing 1% (vol/vol) distilled water, (c) grinding the NaOH beads to a slurry.

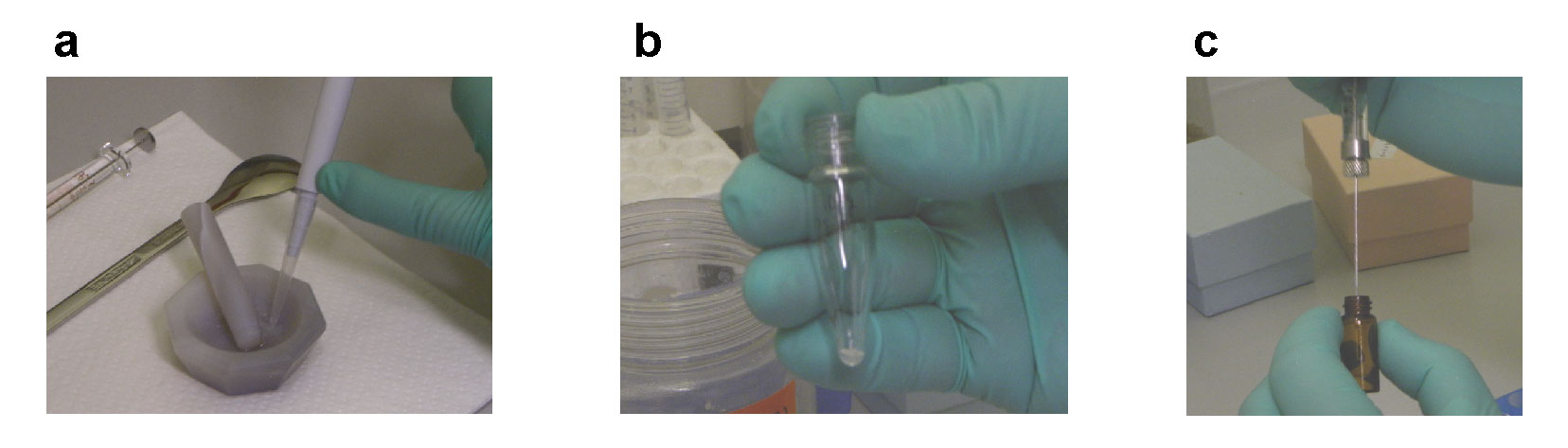
Fig. 2. Transfer the reagents into the reaction tube
(a) measuring off the NaOH slurry using a micropipette, (b) the reaction tube containing the slurry, (c) measuring off methyl iodide using a micro syringe.


Fig. 3. Cutting the end off a pipette tip |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-06-19 13:46:36 |
- Hakomori, S. (1964) A rapid permethylation of glycolipid, and polysaccharide catalyzed by methylsulfinyl carbanion in dimethyl sulfoxide. J. Biochem. 55, 205–208 [PMID : 14135466]
- Ciucanu, I., and Kerek, F. (1984) A simple and rapid method for the permethylation of carbohydrates. Carbohydrate Research 131, 209–217 [PMID : not found]
- Ciucanu, I., and Costello, C. E. (2003) Elimination of oxidative degradation during the per-O-methylation of carbohydrates. J. Am. Chem. Soc. 125, 16213–16219 [PMID : 14692762]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Kameyama, Akihiko,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.30,4,2024 .
How to Cite this Work in Website:
Kameyama, Akihiko,
(2014).
Permethylation for glycan analysis.
Retrieved 30,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t131.
html source
Kameyama, Akihiko,
(2014).
<b>Permethylation for glycan analysis</b>.
Retrieved 4 30,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t131" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t131</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|