There are two major sulfoglycolipids in the mammal: one being the sulfatide, which is a sphingolipid and the other being the seminolipid, which is an ether glycerolipid. Sulfatide is a major lipid component of the myelin sheath and is synthesized in myelin-generating cells, oligodendrocyes in the central nervous system and Schwann cells in the peripheral nervous system. Seminolipid is synthesized in spermatocytes and maintained in the subsequent germ cell stages.
The carbohydrate moiety of sulfatide and seminolipid has the same structure and is biosynthesized via sequential reactions catalyzed by common enzymes: ceramide galactosyltransferase (CGT, EC 2.4.1.45) and cerebroside sulfotransferase (CST, EC 2.8.2.11) (Fig. 1). Cerebroside is the traditional name of galactosylceramide. CST is also known as sulfatide synthase. CST catalyzes the transfer of sulfonate group from the activated sulfate, 3’-phosphoadenosine 5’-phosphosulfate (PAPS) to the C3 position of the nonreducing terminal galactose of glycolipid oligosaccharides (Fig. 2). CST was purified homogeneously from human renal cancer cells where CST is very highly expressed 2). Subsequently a cDNA clone encoding human CST was isolated from a cDNA library of human renal cancer cells, using degenerate oligonucleotides synthesized based on the amino acid sequence data of the purified enzyme 3), followed by cloning of human genomic DNA and mouse cDNA and genomic DNA1). CST that is composed of 423 amino acids has type II transmembrane topology and two PAPS recognizing motifs as other Golgi-resident sulfotransferases do. Two N-glycan oligosaccharide chains are attached to CST, and the C-terminal one is essential for its enzymatic activity. CST is expressed in a tissue-specific manner, and is highly expressed in brain, kidney, testis and alimentary system. This tissue-specific expression of the CST gene is regulated by the alternative usage of multiple promoters1). CST gene is assigned to human chromosome 22q12 and mouse chromosome 11.
CST is the sole sulfotransferase responsible for the biosynthesis of sulfatide, which is rich in myelin sheath, and seminolipid, which is unique in spermatogenic cells. Cst-null mice generated by gene targeting manifest some neurological disorders due to myelin dysfunction and an arrest of spermatogenesis, indicating that CST and its products, or sulfoglycolipids are essential for organisms 4). CST is the first member identified in the βGal 3-O-sulfotransferase family. The other three members, Gal3ST-2~4, have been identified in the EST database by making use of homology to the CST gene. All the Gal3ST-2~4 actually showed sulfotransferase activity in vitro, but act on glycoproteins.
A convenient assay method for CST activity was developed using anion-exchange chromatography 5). The reaction mixture contains 5 nmol of GalCer, 0.5 μmol of MnCl2, 1 nmol of [35S]PAPS (100 cpm/pmol), 0.5 mg of Lubrol PX, 12.5 nmol of dithiothreitol, 0.25 μmol of NaF, 0.1 μmol of ATP, 20 mg of BSA and enzyme protein in 25 mM Na cacodylate-HCl, pH 6.5, in a total volume of 50 ml. After incubation at 37°C for 1 h, the reaction is terminated with 1 mL of chloroform/methanol/water (30:60:8). The reaction product is isolated on a DEAE-Sephadex A-25 column and assayed for radioactivity using a liquid scintillation counter. The values are corrected for a blank value, which is obtained by using a reaction mixture devoid of the acceptor.
The substrate specificity of CST purified from human renal cancer cells was investigated 2). GalCer is the best acceptor and LacCer, galactosyl 1-alkyl-2acyl-sn-glycerol (the precursor for seminolipid), galactosyl diacylglycerol are also good acceptors. GlcCer, Gg3Cer, Gg4Cer, Gb4Cer, and nLc4Cer serve as acceptors although the relative activities are low. On the other hand, the enzyme cannot act on Gb3Cer, which possesses α-galactoside at the nonreducing terminus. Neither galactose nor lactose serves as an acceptor. These observations suggest that CST prefers β-galactoside at the nonreducing termini of sugar chains attached to a lipid moiety. |
| Category | Glycosyltransferases & related proteins |
| Protocol Name | Enzyme assay of sulfotransferase for cerebroside |
Authors
 |
Honke, Koichi
Department of Biochemistry, Kochi University Medical School
|
| KeyWords |
|
Reagents
 |
| ● |
Sodium cacodylate (Wako Pure Chemical Industries Ltd., Osaka, Japan) |
| ● |
GalCer (Sigma-Aldrich, St. Louis, MO) |
| ● |
3’-phospoadenosine-5’-phosphosulfate (PAPS) (Sigma-Aldrich) |
| ● |
[35S]PAPS (NEN/PerkinElmer, Waltham, MA) |
| ● |
MnCl2 (Wako Pure Chemical Industries Ltd.) |
| ● |
Lubrol PX (polyoxyethylene (23) lauryl ether) (Wako Pure Chemical Industries Ltd.) |
| ● |
Dithiothreitol (DTT) (Wako Pure Chemical Industries Ltd.) |
| ● |
NaF (Wako Pure Chemical Industries Ltd.) |
| ● |
ATP (Wako Pure Chemical Industries Ltd.) |
| ● |
NaCl (Wako Pure Chemical Industries Ltd.) |
| ● |
DEAE Sephadex A-25 (GE Healthcare, Little Chalfont, UK) |
| ● |
Chloroform (Wako Pure Chemical Industries Ltd.) |
| ● |
Methanol (Wako Pure Chemical Industries Ltd.) |
| ● |
Scintillation cocktail (PerkinElmer, Waltham, MA) |
|
Instruments
 |
| ● |
|
| ● |
|
| ● |
|
| ● |
Liquid scintillation counter |
|
| Methods |
|
1. |
Enzyme assay of sulfotransferase for cerebroside |
| 1) |
Prepare the reaction mixture in a total volume of 50 μL as follows.
| |
|
|
final concetration |
| 250 mM |
Na cacodylate, pH 6.4 |
5 μL |
25 mM |
| 0.5 mM |
GalCer in 5% TRX-100 |
5 |
50 μM |
| 0.1 M |
MnCl2 |
5 |
10 mM |
| 10% |
Lubrol PX |
5 |
1% |
| 12.5 mM |
DTT |
1 |
0.25 mM |
| 0.25 M |
NaF |
1 |
5 mM |
| 0.1 M |
ATP |
1 |
2 mM |
| 0.5 M |
NaCl |
5 |
50 mM |
| 1 mM |
[35S]PAPS |
2 |
40 μM |
| |
(ca 100 dpm/pmol) |
|
|
|
Comment 0
|

|
| 4) |
Terminate the reaction with 1 mL of chloroform/methanol/water (30:60:8 v/v). |
Comment 0
|

|
| 5) |
Apply the whole reaction product onto a mini column packed with 1 mL of DEAE-Sephadex A-25 resin. |
Comment 0
|

|
| 6) |
Wash with 3 mL of chloroform/methanol/water (30:60:8 v/v). |
Comment 0
|

|
| 8) |
Elute with 5 mL of 90 mM ammonium acetate in methanol. Eluate is directly collected into a scintillation vial. |
Comment 0
|

|
| 9) |
Put a scintillation cocktail into the eluate and count the radioactivity with a liquid scintillation counter. The values are corrected for a blank value, which is obtained using a reaction mixture devoid of GalCer. |
Comment 0
|
|
|
| Notes | Since the half time of 35S is relatively short (87.5 day), specific activity of [35S]PAPS changes day by day. Therefore, the radioactivity of donor substrate must be checked every time when used.
Enzyme activity is calculated by the following formula:
Enzyme activity (pmol/h/mL) = ( sample - blank (dpm)) ÷ specific activity of [35S]PAPS (dpm/pmol) × 50
This method using anion exchange chromatography is applicable to the enzyme assay for ganglioside synthases. |
| Figure & Legends |
Figure & Legends 
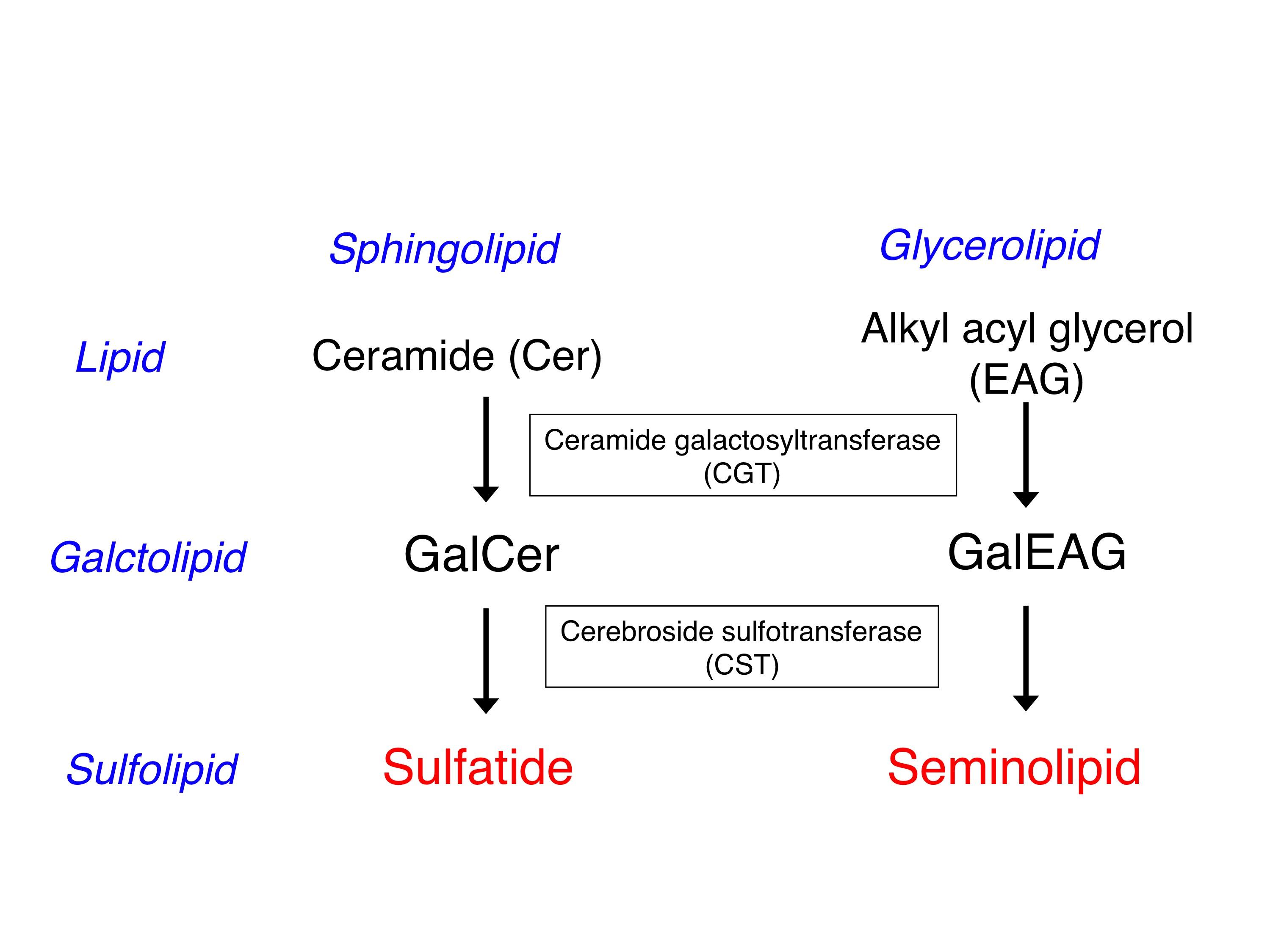
Fig. 1. Biosynthetic pathway of sulfatide and seminolipid

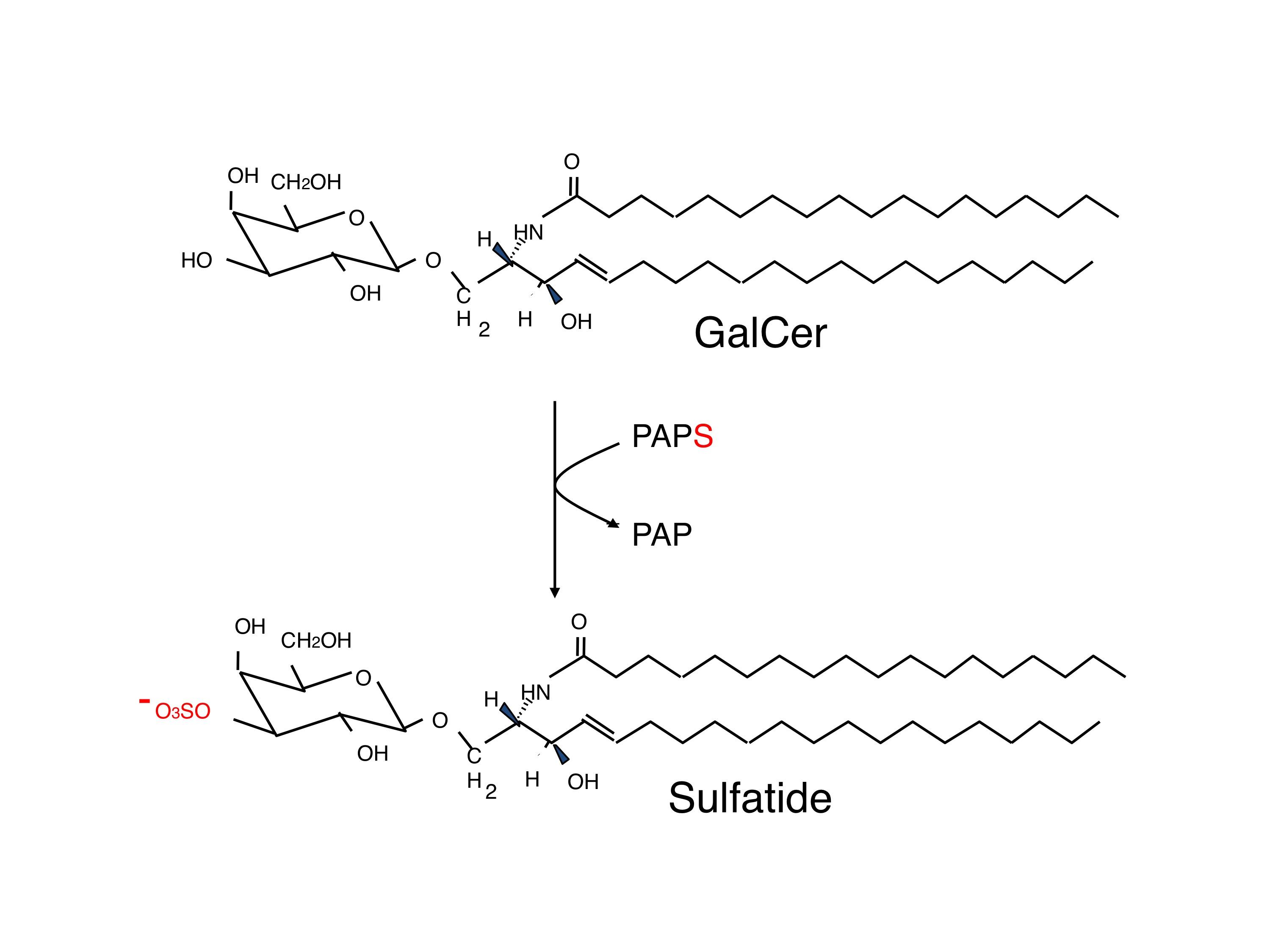
Fig. 2. Reaction mediated by cerebroside sulfotransferase (CST) |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-07-31 09:11:59 |
- Hirahara, Y., Tsuda, M., Wada, Y., and Honke, K. (2000) cDNA cloning, genomic cloning, and tissue-specific regulation of mouse cerebroside sulfotransferase. Eur J Biochem. 267, 1909–17 [PMID : 10727929]
- Honke, K., Yamane, M., Ishii, A., Kobayashi, T., and Makita, A. (1996) Purification and characterization of 3'-phosphoadenosine-5'-phosphosulfate:GalCer sulfotransferase from human renal cancer cells. J Biochem. 119, 421–7 [PMID : 8830034]
- Honke, K., Tsuda, M., Hirahara, Y., Ishii, A., Makita, A., and Wada, Y. (1997) Molecular cloning and expression of cDNA encoding human 3'-phosphoadenylylsulfate:galactosylceramide 3'-sulfotransferase. J Biol Chem. 272, 4864–8 [PMID : 9030544]
- Honke, K., Hirahara, Y., Dupree, J., Suzuki, K., Popko, B., Fukushima, K., Fukushima, J., Nagasawa, T., Yoshida, N., Wada, Y., and Taniguchi, N. (2002) Paranodal junction formation and spermatogenesis require sulfoglycolipids. Proc Natl Acad Sci U S A. 99, 4227–32 [PMID : 11917099]
- Kawano, M., Honke, K., Tachi, M., Gasa, S., and Makita, A. (1989) An assay method for ganglioside synthase using anion-exchange chromatography. Anal Biochem. 182, 9–15 [PMID : 2513740]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Honke, Koichi,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.1,5,2024 .
How to Cite this Work in Website:
Honke, Koichi,
(2014).
Enzyme assay of sulfotransferase for cerebroside.
Retrieved 1,5,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t124.
html source
Honke, Koichi,
(2014).
<b>Enzyme assay of sulfotransferase for cerebroside</b>.
Retrieved 5 1,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t124" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t124</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|