O-linked β-N-acetylglucosaminylation (O-GlcNAcylation) is a dynamic post-translational modification found on the Ser/Thr residue of nucleocytoplasmic proteins. More than 500 proteins have been identified to be O-GlcNAcylated and O-GlcNAcylation is thought to regulate many cellular processes. Removal (hydrolysis) of the O-GlcNAc residues from O-GlcNAcylated proteins is catalyzed by the nucleocytoplasmic O-linked β-N-acetylglucosaminidase (O-GlcNAcase). In this protocol, a standard protocol for pNP-O-GlcNAc-based in vitro O-GlcNAcase enzyme assay is described. A flowchart of this assay is shown in Fig. 1. |
| Category | Biosynthesis & Metabolism |
| Protocol Name | In vitro enzyme assay of nucleocytoplasmic O-linked β-N-acetylglucosaminidase (O-GlcNAcase) |
Authors
 |
Kamemura, Kazuo
Department of Bioscience, Nagahama Institute of Bio-Science & Technology
|
| KeyWords |
|
Reagents
 |
| ● |
p-Nitrophenyl-N-acetyl-β-D-glucosaminide (pNP-O-GlcNAc) (Sigma-Aldrich, St. Louis, MO) |
| ● |
Bovine serum albumin (BSA) |
| ● |
|
|
Instruments
 |
|
| Methods |
|
1. |
In vitro enzyme assay of nucleocytoplasmic O-linked β-N-acetylglucosaminidase (O-GlcNAcase) |
| 1) |
Prepare O-GlcNAcase enzyme fraction. |
Comment 1
|

|
| 2) |
Prepare the reaction mixture (final volume of 100 μL).
50 mM sodium cacodylate (pH 6.5)
2 mM pNP-O-GlcNAc
0.3% BSA
O-GlcNAcase enzyme fraction |
Comment 1
|

|
| 4) |
Stop the reaction by the addition of 900 μL of 500 mM sodium carbonate. |
Comment 0
|

|
| 5) |
Measure the absorbance of the cleaved pNP by spectrophotometer at 400–405 nm. |
Comment 0
|
|
|
| Figure & Legends |
Figure & Legends 
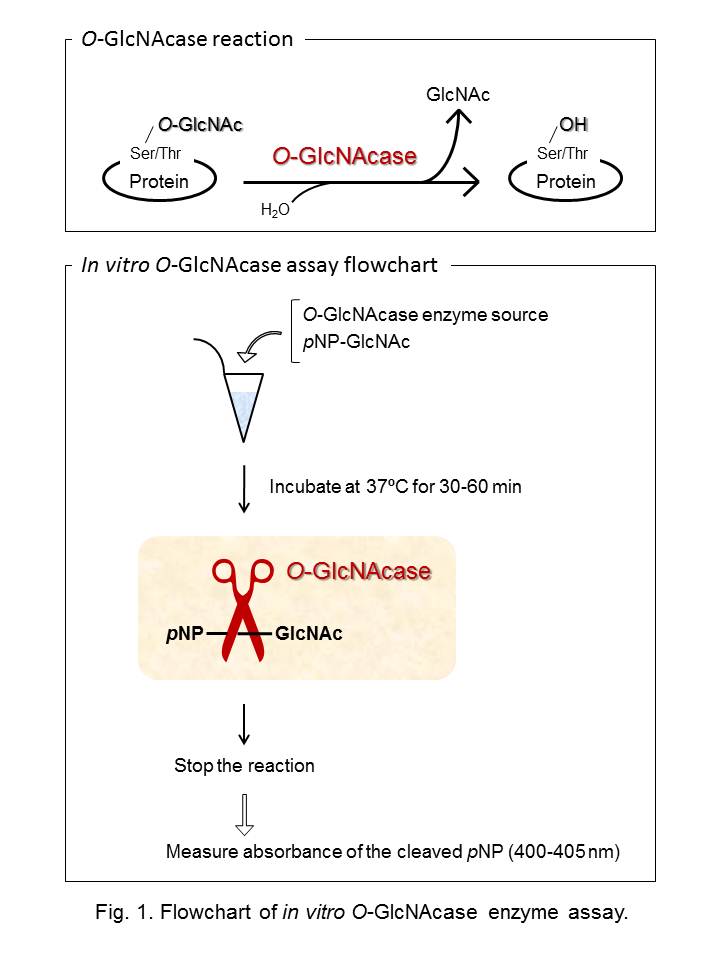
|
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-06-04 10:11:31 |
- Dong, D.L., and Hart, G.W. (1994) Purification and characterization of an O-GlcNAc selective N-acetyl-β-D-glucosaminidase from rat spleen cytosol. J. Biol. Chem. 269, 19321–19330. [PMID : 8034696]
- Gao, Y., Wells, L., Comer, F.I., Parker, G.J., and Hart, G.W. (2001) Dynamic O-glycosylation of nuclear and cytosolic proteins: Cloning and characterization of a neutral cytosolic β-N-acetylglucosaminidase from bovine brain. J. Biol. Chem. 276, 9838–9845. [PMID : 11148210]
- Wells, L., Gao, Y., Mahoney, J.A., Vosseller, K., Chen, C., Rosen, A., and Hart, G.W. (2002) Dynamic O-glycosylation of nuclear and cytosolic proteins: Further characterization of the nucleocytoplasmic β-N-acetylglucosaminidase, O-GlcNAcase. J. Biol. Chem. 277, 1755–1761. [PMID : 11788610]
- Toleman, C., Paterson, A.J., and Kudlow, J.E. (2006) Location and characterization of the O-GlcNAcase active site. Biochim. Biophys. Acta 1760, 829–839. [PMID : 16517082]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Kamemura, Kazuo,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.30,4,2024 .
How to Cite this Work in Website:
Kamemura, Kazuo,
(2014).
In vitro enzyme assay of nucleocytoplasmic O-linked β-N-acetylglucosaminidase (O-GlcNAcase).
Retrieved 30,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t109.
html source
Kamemura, Kazuo,
(2014).
<b><em>In vitro</em> enzyme assay of nucleocytoplasmic <em>O</em>-linked β-<em>N</em>-acetylglucosaminidase (<em>O</em>-GlcNAcase)</b>.
Retrieved 4 30,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t109" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t109</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|