
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002857 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-002857

|
||||
Regist Date |
2012/06/21 19:59:45 | ||||
| REACTANT | |||||
|
|
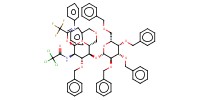
|
||||
Reactant Type |
donor | ||||
Mol |
1.17 mmol | ||||
|
|
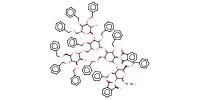
|
||||
Reactant Type |
acceptor | ||||
Mol |
0.34 mmol | ||||
|
|
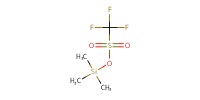
|
||||
Reactant Type |
TMSOTf | ||||
Mol |
30 micro mole | ||||
| PRODUCT | |||||
MOLECULE ID |
|
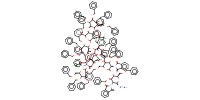
|
|||
Yield |
85% | ||||
| REACTION DETAIL | |||||
Reaction Time |
2 hours | ||||
Reaction Temp |
-40 degree C | ||||
Solvent |
anhydrous CH2Cl2 (30mL) | ||||
Comment |
MS AW300 was included in the solvent. | ||||
| The addition of the TMSOTf was performed after the addition of all the other reactants. | |||||
| COMMENT | |||||
| Keywords: Biantennary, N-glycans, LacNAc, LacdiNAc, SPPS | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000371 | ||||
Issn |
Electronic | ||||
Doi |
10.1021/jo200149d | ||||
PubMed ID |
21612260 | ||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (13): 5229-39. | ||||
Article Title |
Synthesis of biantennary complex-type nonasaccharyl asn building blocks for solid-phase glycopeptide synthesis. | ||||
Author |
Masashi, Hagiwara; Mizuki, Dohi; Yuko, Nakahara; Keiko, Komatsu; Yuya, Asahina; Akiharu, Ueki; Hironobu, Hojo; Yoshiaki, Nakahara; Yukishige, Ito | ||||
Affiliation |
Department of Applied Biochemistry, Institute of Glycoscience, Tokai University, 4-1-1 Kitakaname, Hiratsuka-shi, Kanagawa 259-1292, Japan. | ||||
Reference Id |
REF-0000-000372 | ||||
Source |
J. Org. Chem. 2011, 76, 5229-5239 | ||||
Doi |
10.1021/jo200149d | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|