
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002664 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-002664

|
|||||||
Regist Date |
2012/06/21 19:36:16 | |||||||
| REACTANT | ||||||||
|
|
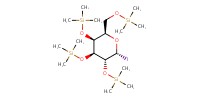
|
|||||||
Reactant Type |
(in CH2Cl2) | |||||||
Mol |
less than 10.5 mmol | |||||||
|
|
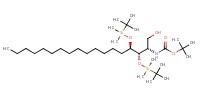
|
|||||||
Mol |
3.50 mmol | |||||||
|
|
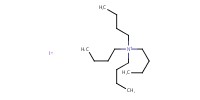
|
|||||||
Reactant Type |
TBAI | |||||||
Mol |
21.0 mmol | |||||||
|
|
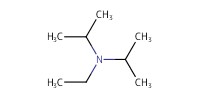
|
|||||||
Reactant Type |
DIEA | |||||||
Mol |
15.8 mmol | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
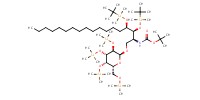
|
|
|||||
Yield |
75%(at least) | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
overnight | |||||||
Reaction Temp |
room temp | |||||||
Solvent |
CH2Cl2 (40mL) | |||||||
Comment |
16, TBAI, DIEA, and MS 4A were mixed and stirred for 15 minutes at room temperature before the reaction. | |||||||
| MS 4A was included in the solvent. | ||||||||
| 15 was added dropwise over 15 minutes. | ||||||||
| COMMENT | ||||||||
| Keywords: building block, ceramides, glycolipids, galactosyl-phytosphingosine, KRN7000 | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000361 | |||||||
Issn |
Electronic | |||||||
Doi |
10.1021/jo102064p | |||||||
PubMed ID |
21155575 | |||||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (1): 320-3. | |||||||
Article Title |
Synthesis of a versatile building block for the preparation of 6-N-derivatized α-galactosyl ceramides: rapid access to biologically active glycolipids. | |||||||
Author |
Peter J, Jervis; Liam R, Cox; Gurdyal S, Besra | |||||||
Affiliation |
School of Biosciences, University of Birmingham, Edgbaston, Birmingham B15 2TT, United Kingdom. | |||||||
Reference Id |
REF-0000-000362 | |||||||
Source |
J. Org. Chem. 2011, 76, 320-323 | |||||||
Doi |
10.1021/jo102064p | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|