
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002593 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-002593

|
|||||||
Regist Date |
2012/06/21 19:29:34 | |||||||
| REACTANT | ||||||||
|
|
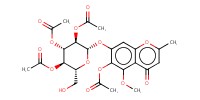
|
|
||||||
Mol |
0.13 mmol | |||||||
|
|
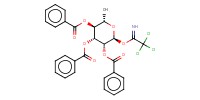
|
|||||||
Mol |
0.19 mmol | |||||||
|
|
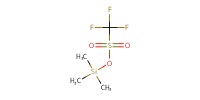
|
|||||||
Reactant Type |
TMSOTf | |||||||
Mol |
1 + 1 equivalent | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
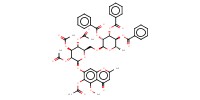
|
|
|||||
Yield |
60% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
2 hours, 3 hours | |||||||
Reaction Temp |
-20 to 0 degree C, 0 degree C | |||||||
Solvent |
anhydrous CH2Cl2 (6.0mL), anhydrous CH2Cl2 (6.0mL) | |||||||
Comment |
1) 8+9a, MS 4A, TMSOTf, 2) second addition of TMSOTf | |||||||
| MS 4A was included in the solvent. | ||||||||
| 8, 9a, and MS 4A were mixed and stirred for 1 hour at room temperature before the reaction. | ||||||||
| The reactants were mixed at -20 degree in Celsius, and allowed to warm to 0 degree within the reaction time. (first phase) | ||||||||
| COMMENT | ||||||||
| Keywords: 2-methylchromone-7-O-rutinosides, cytotoxicity, human tumor cell lines, acidic labile, 2-methylchromone aglycon | ||||||||
| There are multiple phases in this reaction. | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000355 | |||||||
Issn |
Electronic | |||||||
Doi |
10.1021/jo102325s | |||||||
PubMed ID |
21366286 | |||||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (7): 2265-8. | |||||||
Article Title |
Concise synthesis of 5-methoxy-6-hydroxy-2-methylchromone-7-O- and 5-hydroxy-2-methylchromone-7-O-rutinosides. Investigation of their cytotoxic activities against several human tumor cell lines. | |||||||
Author |
Baolin, Wu; Wenpeng, Zhang; Zhonghua, Li; Li, Gu; Xin, Wang; Peng George, Wang | |||||||
Affiliation |
Department of Chemistry, The Ohio State University, 484 West 12th Avenue, Columbus, Ohio 43210, USA. | |||||||
Reference Id |
REF-0000-000356 | |||||||
Source |
J. Org. Chem. 2011, 76, 2265-2268 | |||||||
Doi |
10.1021/jo102325s | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|