
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002460 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-002460

|
|||||||
Regist Date |
2012/06/21 19:16:10 | |||||||
| REACTANT | ||||||||
|
|
|
|
||||||
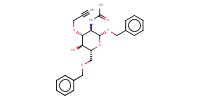
Reactant Type |
(in 45 mL of CH2Cl2) | |||||||
Mol |
0.284 mmol | |||||||
|
|
|
|||||||
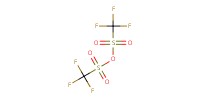
Reactant Type |
Tf2O | |||||||
Mol |
0.568 mmol | |||||||
|
|
|
|||||||
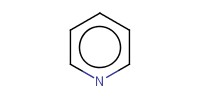
Reactant Type |
pyridine (diluted with 1 mL of CH2Cl2) | |||||||
Volume |
0.09 mL | |||||||
|
|
|
|||||||
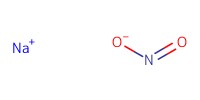
Reactant Type |
NaNO2 | |||||||
Mol |
2.84 mmol | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
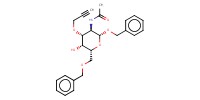
|
|
|||||
Yield |
40% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
30 minutes, 1 hour | |||||||
Reaction Temp |
-18 degree C, room temp | |||||||
Solvent |
CH2Cl2 (10mL), DMF (12mL) | |||||||
Comment |
1) 27+Tf2O, pyridine, 2) +NaNO2 | |||||||
| Pyridine was added dropwise. (first phase) | ||||||||
| The addition of 27 was performed after the addition of all the other reactants. (first phase) | ||||||||
| COMMENT | ||||||||
| Keywords: triazole, heparosan, chondroitin, copper catalyzed azide-alkyne cycloaddition, CuAAC, azido-glucuronic acid, glucosamine, galactosamine | ||||||||
| There are multiple phases in this reaction. | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000343 | |||||||
Issn |
Electronic | |||||||
Doi |
10.1021/jo200076z | |||||||
PubMed ID |
21438620 | |||||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (9): 3181-93. | |||||||
Article Title |
Design and synthesis of unnatural heparosan and chondroitin building blocks. | |||||||
Author |
Smritilekha, Bera; Robert J, Linhardt | |||||||
Affiliation |
Department of Chemistry and Chemical Biology, Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, New York 12180, USA. | |||||||
Reference Id |
REF-0000-000344 | |||||||
Source |
J. Org. Chem. 2011, 76, 3181-3193 | |||||||
Doi |
10.1021/jo200076z | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|