
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002312 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-002312

|
||||
Regist Date |
2012/06/21 19:01:25 | ||||
| REACTANT | |||||
|
|
|
||||
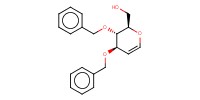
Reactant Type |
sugar (1,5-anhydro-3,4-di-O-benzyl-2-deoxy-D-arabino-hex-1-enuronate) | ||||
Mol |
3.06 mmol | ||||
|
|
|
||||
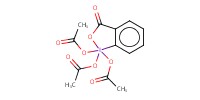
Reactant Type |
DMP | ||||
Mol |
3.68 mmol | ||||
|
|
|
||||
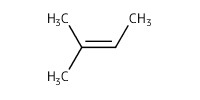
Reactant Type |
2-methyl-2-butene | ||||
Mol |
5.09 mmol | ||||
|
|
|
||||
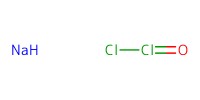
Reactant Type |
NaOCl2 (in H2O) | ||||
Mol |
6.79 mmol | ||||
|
|
|
||||
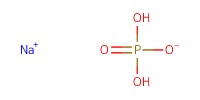
Reactant Type |
NaH2PO4 (in H2O) | ||||
Mol |
5.09 mmol | ||||
|
|
|
||||
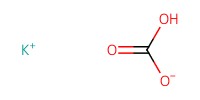
Reactant Type |
KHCO3 | ||||
Mol |
4.25 mmol | ||||
|
|
|
||||
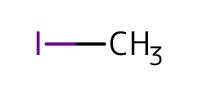
Reactant Type |
MeI | ||||
Mol |
4.18 mmol | ||||
| PRODUCT | |||||
MOLECULE ID |
|
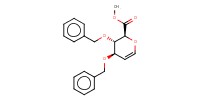
|
|||
Yield |
77% | ||||
| REACTION DETAIL | |||||
Reaction Time |
2 hours, 2 hours, 3 hours | ||||
Reaction Temp |
room temp, room temp, room temp | ||||
Solvent |
anhydrous CH2Cl2, t-BuOH, anhydrous DMF | ||||
Comment |
1) sugar+DMP, 2) +2-methyl-2-butene, NaOCl2, NaH2PO4, 3) +KHCO3, MeI | ||||
| COMMENT | |||||
| Keywords: 2-acylamino uronic acid, glycosyl phosphonates, oxidation, uronate 2-nitro-glycal, thermodynamic 1,2-trans-di-equatorial phosphonylation | |||||
| There are multiple phases in this reaction. | |||||
| ATTENTION: The chemical structure of NaOCl2 could not be found. (a plausible structure is being registered) | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000331 | ||||
Issn |
Electronic | ||||
Doi |
10.1021/jo2002193 | ||||
PubMed ID |
21495699 | ||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (10): 4099-104. | ||||
Article Title |
Accessing C-1 phosphonylated 2-acylamino uronic acids via 2-nitro-glycals. | ||||
Author |
Beenu, Bhatt; Robin J, Thomson; Mark, von Itzstein | ||||
Affiliation |
Institute for Glycomics, Griffith University, Gold Coast Campus, Queensland 4222, Australia. | ||||
Reference Id |
REF-0000-000332 | ||||
Source |
J. Org. Chem. 2011, 76, 4099-4104 | ||||
Doi |
10.1021/jo2002193 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|