
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0002243 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-002243

|
||||
Regist Date |
2012/06/21 18:53:43 | ||||
| REACTANT | |||||
|
|
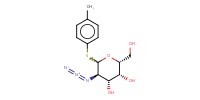
|
||||
Mol |
2.56 mmol | ||||
|
|
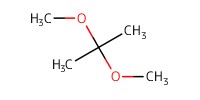
|
||||
Reactant Type |
Me2C(OMe)2 (solvent) | ||||
Volume |
20 mL | ||||
|
|
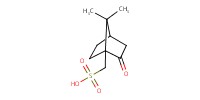
|
||||
Reactant Type |
CSA | ||||
Mol |
catalytic amount | ||||
| PRODUCT | |||||
MOLECULE ID |
|
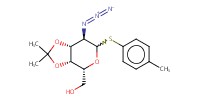
|
|||
Yield |
80% | ||||
| REACTION DETAIL | |||||
Reaction Time |
24 hours, NOT specified | ||||
Reaction Temp |
room temp, reflux | ||||
Solvent |
Me2C(OMe)2, MeOH/H2O = 10/1 | ||||
Comment |
1) +all, 2) temperature change, solvent change | ||||
| COMMENT | |||||
| Keywords: stereoselectivity, 2-azido-2-deoxygalactosyl donors, acetyl groups, temperature, computational methods | |||||
| There are multiple phases in this reaction. | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000327 | ||||
Issn |
Electronic | ||||
Doi |
10.1021/jo1025157 | ||||
PubMed ID |
21574599 | ||||
Journal Name |
The Journal of organic chemistry. (2011) 76 (13): 5207-18. | ||||
Article Title |
Study of the stereoselectivity of 2-azido-2-deoxygalactosyl donors: remote protecting group effects and temperature dependency. | ||||
Author |
Jane, Kalikanda; Zhitao, Li | ||||
Affiliation |
Department of Chemistry, Binghamton University, Binghamton, New York 13902, United States. | ||||
Reference Id |
REF-0000-000328 | ||||
Source |
J. Org. Chem. 2011, 76, 5207-5218 | ||||
Doi |
10.1021/jo1025157 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|