
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001754 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-001754

|
|||||||
Regist Date |
2012/06/21 17:51:34 | |||||||
| REACTANT | ||||||||
|
|
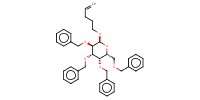
|
|
||||||
Mol |
1.3 mmol | |||||||
|
|
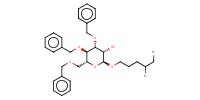
|
|
||||||
Mol |
1.0 mmol | |||||||
|
|
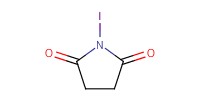
|
|||||||
Reactant Type |
NIS | |||||||
Mol |
1.56 mmol | |||||||
|
|
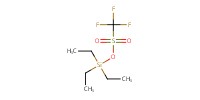
|
|||||||
Reactant Type |
TESOTf | |||||||
Mol |
0.26 mmol | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
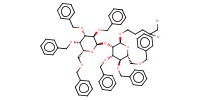
|
|
|||||
Product Type |
alpha | |||||||
Yield |
98%(alpha/beta=11/1) | |||||||
MOLECULE ID |
|
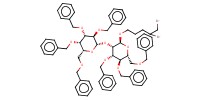
|
||||||
Product Type |
beta | |||||||
Yield |
98%(alpha/beta=11/1) | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
10 minutes | |||||||
Reaction Temp |
room temp | |||||||
Solvent |
dry CH2Cl2 | |||||||
Comment |
The addition of NIS was performed before the addition of TESOTf. | |||||||
| TESOTf was added dropwise. | ||||||||
| COMMENT | ||||||||
| Keywords: oligosaccharide synthesis, n-pentenyl glycosides, NPG Coupling, NIS-Catalytic Et3SiOTf | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000196 | |||||||
Source |
PREPARATIVE CARBOHYDRATE CHEMISTRY | |||||||
Reference Id |
REF-0000-000197 | |||||||
Source |
ISBN 0-8247-9802-3 | |||||||
Reference Id |
REF-0000-000237 | |||||||
Source |
J. Am. Chem. Soc. 59:4443 (1994) | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|