
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001706 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-001706

|
|||||||
Regist Date |
2012/06/21 17:40:37 | |||||||
| REACTANT | ||||||||
|
|
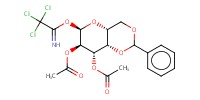
|
|
||||||
Mol |
32 mmol | |||||||
|
|
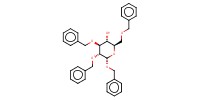
|
|
||||||
Mol |
20 mmol | |||||||
|
|
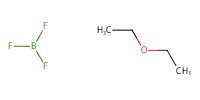
|
|||||||
Reactant Type |
BF3*OEt2 | |||||||
Mol |
32.5 mmol | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
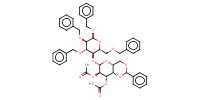
|
|
|||||
Yield |
84% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
30 minutes | |||||||
Reaction Temp |
0 degree C | |||||||
Solvent |
dry n-hexane | |||||||
| COMMENT | ||||||||
| Keywords: glycosylation, O-glycosyl trichloroacetimidates | ||||||||
| ATTENTION: CH2Cl2(solvent) appearing in the scheme is not mentioned in the written method. | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000196 | |||||||
Source |
PREPARATIVE CARBOHYDRATE CHEMISTRY | |||||||
Reference Id |
REF-0000-000197 | |||||||
Source |
ISBN 0-8247-9802-3 | |||||||
Reference Id |
REF-0000-000209 | |||||||
Source |
T. Stauch, Dissertation, Universitat Konstanz, 1995 | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|