
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001608 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-001608

|
||||
Regist Date |
2012/06/21 17:25:34 | ||||
| REACTANT | |||||
|
|
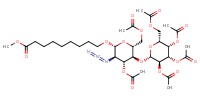
|
||||
Mol |
0.632 mmol | ||||
|
|
|
||||
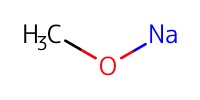
Reactant Type |
NaOMe (0.5 M methanolic solution) | ||||
Volume |
0.27 mL | ||||
|
|

|
||||
Reactant Type |
FeCl3 (in acetone) | ||||
Mol |
0.11 mmol | ||||
| PRODUCT | |||||
MOLECULE ID |
|
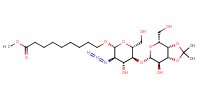
|
|||
Yield |
40% | ||||
MOLECULE ID |
|
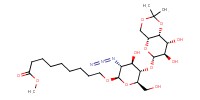
|
|||
Product Type |
isomer | ||||
Yield |
18% | ||||
| REACTION DETAIL | |||||
Reaction Time |
overnight, 30 minutes | ||||
Reaction Temp |
room temp, reflux | ||||
Solvent |
MeOH, acetone | ||||
Comment |
1) 3+NaOMe, 2) +FeCl3 | ||||
| COMMENT | |||||
| Keywords: biological activity, carbohydrates, conformation analysis, lectins, synthesis design | |||||
| There are multiple phases in this reaction. | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000189 | ||||
Issn |
|||||
Doi |
10.1002/chem.200400831 | ||||
PubMed ID |
15770712 | ||||
Journal Name |
Chemistry (Weinheim an der Bergstrasse, Germany). (2005) 11 (10): 3032-8. | ||||
Article Title |
Synthesis and evaluation of 5-thio-L-fucose-containing oligosaccharide. | ||||
Author |
Masayuki, Izumi; Osamu, Tsuruta; Yasuhiro, Kajihara; Shin, Yazawa; Hideya, Yuasa; Hironobu, Hashimoto | ||||
Affiliation |
Department of Life Science, Graduate school of Bioscience and Biotechnology, Tokyo Institute of Technology, 4259 Nagatsuta, Yokohama 226-8501, Japan.masayuki.izumi@aist.go.jp | ||||
Reference Id |
REF-0000-000190 | ||||
Source |
Chem. Eur. J. 2005, 11, 3032-3038 | ||||
Doi |
10.1002/chem.200400831 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|