
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001573 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-001573

|
||||
Regist Date |
2012/06/21 17:22:28 | ||||
| REACTANT | |||||
|
|
|
||||
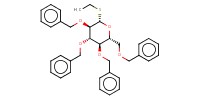
Reactant Type |
glucopyranoside | ||||
Mol |
3.75 mmol | ||||
|
|
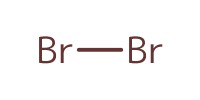
|
||||
Reactant Type |
Br2 (in CH2Cl2, 1/165 in volume) | ||||
Volume |
36 mL | ||||
|
|
|
||||
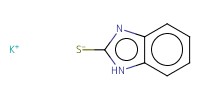
Reactant Type |
KSBiz | ||||
Mol |
9.36 mmol | ||||
|
|
|
||||
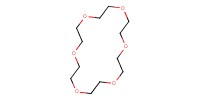
Reactant Type |
18-crown-6 | ||||
Mol |
0.75 mmol | ||||
| PRODUCT | |||||
MOLECULE ID |
|
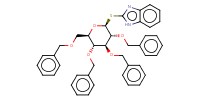
|
|||
Yield |
73% | ||||
| REACTION DETAIL | |||||
Reaction Time |
5 minutes, 16 hours | ||||
Reaction Temp |
room temp, room temp | ||||
Solvent |
CH2Cl2, MeCN | ||||
Comment |
1) glucopyranoside+Br2, MS 3A, 2) +all the rest | ||||
| The glucopyranoside and MS 3A were mixed and stirred for 1 hour at room temperature before the reaction. | |||||
| COMMENT | |||||
| Keywords: carbohydrates, glycosides, glycosylation, oligosaccharides, synthetic methods | |||||
| There are multiple phases in this reaction. | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000187 | ||||
Issn |
Electronic | ||||
Doi |
10.1002/anie.201007212 | ||||
PubMed ID |
21433229 | ||||
Journal Name |
Angewandte Chemie (International ed. in English). (2011) 50 (18): 4197-201. | ||||
Article Title |
S-Benzimidazolyl glycosides as a platform for oligosaccharide synthesis by an active-latent strategy. | ||||
Author |
Scott J, Hasty; Matthew A, Kleine; Alexei V, Demchenko | ||||
Affiliation |
Department of Chemistry and Biochemistry, University of Missouri-St.?Louis, One University Boulevard, St.?Louis, MO 63121, USA. | ||||
Reference Id |
REF-0000-000188 | ||||
Source |
Angew. Chem. Int. Ed. 2011, 50, 4197-4201 | ||||
Doi |
10.1002/anie.201007212 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|