
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001558 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-001558

|
|||||||
Regist Date |
2012/06/21 17:21:02 | |||||||
| REACTANT | ||||||||
|
|
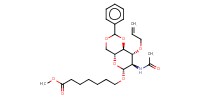
|
|
||||||
Mol |
5 mmol | |||||||
|
|
|
|||||||
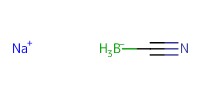
Reactant Type |
NaBH3CN | |||||||
Mol |
45 mmol | |||||||
|
|

|
|||||||
Reactant Type |
HCl (in ether) | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
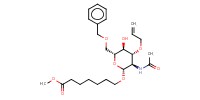
|
|
|||||
Yield |
87% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
0.5 hour (approximately) | |||||||
Reaction Temp |
0 degree C | |||||||
Solvent |
THF | |||||||
Comment |
MS 3A was included in the solvent. | |||||||
| The addition of HCl was continued until the reaction mixture became acidic. | ||||||||
| COMMENT | ||||||||
| Keywords: protection of sugar hydroxy groups, cycloacetal group protection | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000073 | |||||||
Source |
第4版 実験化学講座 (26), 丸善 | |||||||
Reference Id |
REF-0000-000074 | |||||||
Source |
ISBN 4-621-03702-1 | |||||||
Reference Id |
REF-0000-000186 | |||||||
Source |
D. P. Khare, O. Hindsgaul, and R. U. Lemieux, Carbohydr. Res., 136, 285 (1985) | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|