
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0001078 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-001085

|
||||
Regist Date |
2012/06/21 16:38:22 | ||||
| REACTANT | |||||
|
|
|
||||
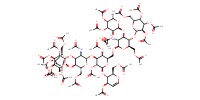
Reactant Type |
peracetylated octasaccharide | ||||
Mol |
0.00500 mmol | ||||
|
|
|
||||
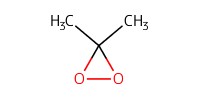
Reactant Type |
DMDO (0.075 M in acetone) | ||||
Volume |
0.15 mL | ||||
|
|
|
||||
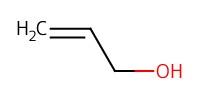
Reactant Type |
allyl alcohol (solvent) | ||||
Volume |
3 mL | ||||
| PRODUCT | |||||
MOLECULE ID |
|
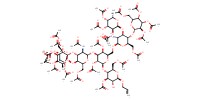
|
|||
Yield |
48% | ||||
MOLECULE ID |
|
|
|||
Product Type |
mannose type isomer | ||||
Yield |
37% | ||||
| REACTION DETAIL | |||||
Reaction Time |
45 minutes, overnight | ||||
Reaction Temp |
0 degree C, room temp | ||||
Solvent |
CH2Cl2, allyl alcohol | ||||
Comment |
1) 59+DMDO, 2) +all the rest | ||||
| COMMENT | |||||
| There are two phases in this reaction. | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000048 | ||||
Issn |
|||||
PubMed ID |
11273599 | ||||
Journal Name |
Journal of the American Chemical Society. (2001) 123 (1): 35-48. | ||||
Article Title |
Total syntheses of tumor-related antigens N3: probing the feasibility limits of the glycal assembly method. | ||||
Author |
H M, Kim; I J, Kim; S J, Danishefsky | ||||
Affiliation |
Laboratory for Bioorganic Chemistry, Sloan-Kettering Institute for Cancer Research, 1275 York Ave., New York, New York 10021, USA. | ||||
Reference Id |
REF-0000-000049 | ||||
Source |
J. Am. Chem. Soc. 2001, 123, 35-48 | ||||
Doi |
10.1021/ja0022730 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|