
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0000948 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-000948

|
|||||||
Regist Date |
2012/06/21 16:31:20 | |||||||
| REACTANT | ||||||||
|
|
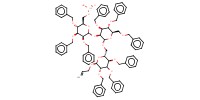
|
|
||||||
Mol |
0.0328 mmol | |||||||
|
|
|
|||||||
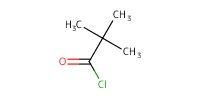
Reactant Type |
PivCl | |||||||
Mol |
0.197 mmol | |||||||
|
|
|
|||||||
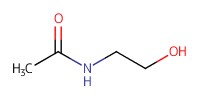
Reactant Type |
AcHNCH2CH2OH | |||||||
Mol |
0.109 mmol | |||||||
|
|

|
|||||||
Reactant Type |
I2 | |||||||
Mol |
0.036 mmol | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
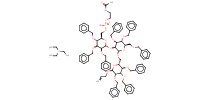
|
||||||
Yield |
58% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
20 minutes + 2 hours, overnight | |||||||
Reaction Temp |
-20 degree C, room temp | |||||||
Solvent |
pyridine, pyridine | |||||||
Comment |
1) 16+PivCl, AcHNCH2CH2OH, 2) +I2 | |||||||
| PivCl was added dropwise for 20 minutes | ||||||||
| COMMENT | ||||||||
| The PMID could not be found. | ||||||||
| There are two phases in this reaction. | ||||||||
| ATTENTION: There are typos in the written method. (compound 6 should be 16, compound 2 should be 17) | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000043 | |||||||
Source |
HETEROCYCLES, Vol. 52, No. 2, 2000, pp. 921-928 | |||||||
Doi |
10.3987/COM-99-S65 | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|