
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0000758 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-000761

|
|||||||
Regist Date |
2012/06/21 16:22:16 | |||||||
| REACTANT | ||||||||
|
|
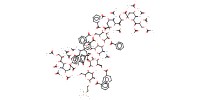
|
|||||||
Mol |
0.0121 mmol | |||||||
|
|
|
|||||||
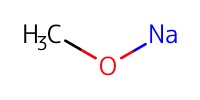
Reactant Type |
NaOMe (28% in MeOH) | |||||||
Mol |
catalytic amounts | |||||||
|
|
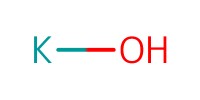
|
|||||||
Reactant Type |
KOH (0.2 M) | |||||||
Volume |
0.5 mL | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
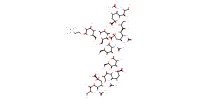
|
|
|||||
Yield |
87% | |||||||
| REACTION DETAIL | ||||||||
Reaction Time |
17 hours, 17 hours, 30 hours | |||||||
Reaction Temp |
room temp, 50 degree C, 75 degree C | |||||||
Solvent |
MeOH, MeOH, MeOH | |||||||
Comment |
1) 45+NaOMe, 2) temperature change, 3) +KOH | |||||||
| COMMENT | ||||||||
| There are three phases in this reaction. | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000035 | |||||||
Issn |
Electronic | |||||||
Doi |
10.1021/jo8027888 | |||||||
PubMed ID |
19296672 | |||||||
Journal Name |
The Journal of organic chemistry. (2009) 74 (8): 3009-23. | |||||||
Article Title |
Ganglioside GQ1b: efficient total synthesis and the expansion to synthetic derivatives to elucidate its biological roles. | |||||||
Author |
Akihiro, Imamura; Hiromune, Ando; Hideharu, Ishida; Makoto, Kiso | |||||||
Affiliation |
Department of Applied Bioorganic Chemistry, Faculty of Applied Biological Sciences, Gifu University, 1-1 Yanagido, Gifu-shi, Gifu 501-1193, Japan. aimamura@ualberta.ca | |||||||
Reference Id |
REF-0000-000036 | |||||||
Source |
J. Org. Chem. 2009, 74, 3009-3023 | |||||||
Doi |
10.1021/jo8027888 | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|