
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0000609 | ||||
Submitter |
The Noguchi Institute | ||||
Reaction ID |
R-0000-000609

|
||||
Regist Date |
2012/06/21 16:14:54 | ||||
| REACTANT | |||||
|
|
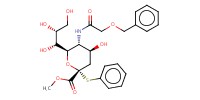
|
||||
Mol |
12.30 mmol | ||||
|
|
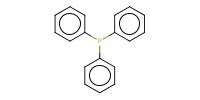
|
||||
Reactant Type |
Ph3P | ||||
Mol |
13.66 mmol | ||||
|
|
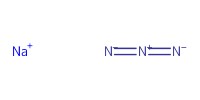
|
||||
Reactant Type |
NaN3 | ||||
Mol |
61.54 mmol | ||||
|
|
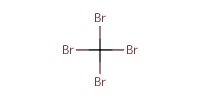
|
||||
Reactant Type |
CBr4 | ||||
Mol |
13.66 mmol | ||||
| PRODUCT | |||||
MOLECULE ID |
|
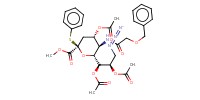
|
|||
Yield |
83.5% | ||||
| REACTION DETAIL | |||||
Reaction Time |
24+3 hours, 6 hours | ||||
Reaction Temp |
room temp, room temp | ||||
Solvent |
dry DMF, Ac2O/pyridine = 20mL/20mL | ||||
Comment |
1) 4+Ph3P, NaN3, CBr4, MeOH, 2) +Ac2O, pyridine | ||||
| MeOH was added 24 hours after the initiation of the first phase. | |||||
| COMMENT | |||||
| There are two phases in this reaction. | |||||
| REFERENCE | |||||
Reference Id |
REF-0000-000028 | ||||
Issn |
Electronic | ||||
Doi |
10.1021/jm8000696 | ||||
PubMed ID |
18841881 | ||||
Journal Name |
Journal of medicinal chemistry. (2008) 51 (21): 6665-81. | ||||
Article Title |
Design, synthesis, and structure-affinity relationships of novel series of sialosides as CD22-specific inhibitors. | ||||
Author |
Hajjaj H M, Abdu-Allah; Taichi, Tamanaka; Jie, Yu; Lu, Zhuoyuan; Magesh, Sadagopan; Takahiro, Adachi; Takeshi, Tsubata; Soerge, Kelm; Hideharu, Ishida; Makoto, Kiso | ||||
Affiliation |
Department of Applied Bio-organic Chemistry, The United Graduate School of Agricultural Sciences, Gifu University, Gifu 501-1193, Japan. | ||||
Reference Id |
REF-0000-000029 | ||||
Source |
J. Med. Chem., 2008, 51, 6665-6681 | ||||
Doi |
10.1021/jm8000696 | ||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|