
|
Glycan Syntheses |
JCGG ID |
JCGG-RAC0000489 | |||||||
Submitter |
The Noguchi Institute | |||||||
Reaction ID |
R-0000-000490

|
|||||||
Regist Date |
2012/06/21 16:09:03 | |||||||
| REACTANT | ||||||||
|
|
|
|
||||||
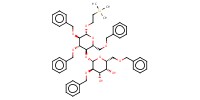
Reactant Type |
lactose acceptor | |||||||
|
|
|
|||||||
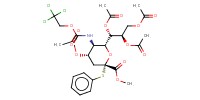
Reactant Type |
sialyl donor | |||||||
|
|
|
|||||||
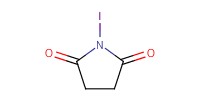
Reactant Type |
NIS | |||||||
|
|

|
|||||||
Reactant Type |
TfOH | |||||||
| PRODUCT | ||||||||
MOLECULE ID |
|
|
|
|||||
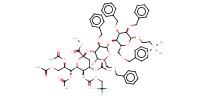
Product Type |
alpha | |||||||
Yield |
83%(alpha/beta=69/14) | |||||||
MOLECULE ID |
|
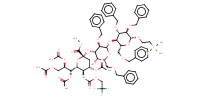
|
|
|||||
Product Type |
beta | |||||||
Yield |
83%(alpha/beta=69/14) | |||||||
| REACTION DETAIL | ||||||||
Reaction Temp |
-50 degree C | |||||||
Solvent |
EtCN-CH2Cl2 (10:1) | |||||||
Catalyst |
NIS, TfOH | |||||||
Comment |
Very few were described regarding this reaction. | |||||||
| COMMENT | ||||||||
| The details regarding this reaction are described in another paper in References. | ||||||||
| REFERENCE | ||||||||
Reference Id |
REF-0000-000021 | |||||||
Issn |
Electronic | |||||||
Doi |
10.1016/j.carres.2009.06.009 | |||||||
PubMed ID |
19560125 | |||||||
Journal Name |
Carbohydrate research. (2009) 344 (12): 1453-63. | |||||||
Article Title |
Study on systematizing the synthesis of the a-series ganglioside glycans GT1a, GD1a, and GM1 using the newly developed N-Troc-protected GM3 and GalN intermediates. | |||||||
Author |
Tatsuya, Komori; Akihiro, Imamura; Hiromune, Ando; Hideharu, Ishida; Makoto, Kiso | |||||||
Affiliation |
Department of Applied Bioorganic Chemistry, Faculty of Applied Biological Sciences, Gifu University, 1-1 Yanagido, Gifu-shi, Gifu 501-1193, Japan. | |||||||
Reference Id |
REF-0000-000022 | |||||||
Source |
Carbohydrate Research, 344 (2009), 1453-1463 | |||||||
Doi |
10.1016/j.carres.2009.06.009 | |||||||
©2008-2013 Research Center for Medical Glycoscience, National Institute of Advanced Industrial Science and Technology (AIST)
|