Endoglycosidases cleave at defined sites within an oligosaccharide chain of glycoproteins/glycolipids. They are the most easy-to-use enzymes for elucidating the function and structure of oligosaccharides, because they can separate both intact oligosaccharide chains and proteins/lipids from glycoconjugates under mild conditions without causing damage.
Endo-β-N-acetylglucosaminidase (EC 3.2.1.96) catalyzes the hydrolysis of the N, N’-diacetylchitobiose moiety in the core region of asparagine-linked oligosaccharides of various glycoproteins. The enzyme has a characteristic to remain one N-acetyl-D-glucosamine residue on the protein. On the other hand, the deglycosylation method using PNGase F cannot remove oligosaccharides unless the protein is denatured. Thus, only endo-β-N-acetylglucosaminidases can be used for deglycosylation of native glycoproteins.
Endo-β-N-acetylglucosaminidase F (Endoglycosidase F or Endo-F) preparations have been divided into three distinct groups, designated Endo-F1, Endo-F2, and Endo-F3 1). Endo-F1 cleaves high-mannose and hybrid-type oligosaccharides, but does not cleave complex-type oligosaccharides (Fig.1)2). Endo-F1 also cleaves core fucosylated hybrid-type structures. Its substrate specificity is similar to that of Endo-H, and it is commonly used as a tool for releasing oligosaccharides from glycoproteins3). Endo-F2 cleaves high-mannose and bi-antennary complex-type oligosaccharides, but does not cleave hybrid-type oligosaccharides (Fig.1)1). Endo-F3 cleaves bi-antennary and tri-antennary complex-type oligosaccharides, but does not cleave high-mannose and hybrid-type oligosaccharides. Endo-F3 also cleaves core fucosyl bi-antennary complex-type and fucosyl tri-mannose core structures (Fig.1)1). |
| Category | N-Glycans |
| Protocol Name | Endo-β-N-acetylglucosaminidase F digestion (Endo-F) |
Authors
 |
Fujita, Kiyotaka
Faculty of Agriculture, Kagoshima University
Yamamoto, Kenji
*
Research Institute for Bioresources and Biotechnology, Ishikawa Prefectural University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Endo-β-N-acetylglucosaminidase F1 (Endoglycosidase F1 or Endo-F1) from Elizabethkingia meningosepticum (formerly known as Chryseobacterium meningosepticum or Flavobacterium meningosepticum).
- Recombinant enzymes (expressed in E. coli) are commercially available from Sigma-Aldrich (St. Louis, MO), QA-Bio (San Mateo, CA), and Merck KGaA (Darmstadt, Germany).
|
| ● |
Endo-β-N-acetylglucosaminidase F2 (Endoglycosidase F2 or Endo-F2) from Elizabethkingia meningosepticum.
- Recombinant enzymes (expressed in E. coli) are commercially available from Sigma-Aldrich, QA-Bio, AMS Biotechnology (Witney, UK), Prozyme Inc. (San Leandro, CA), and Merck KGaA.
|
| ● |
Endo-β-N-acetylglucosaminidase F3 (Endoglycosidase F3 or Endo-F3) from Elizabethkingia meningosepticum.
- Recombinant enzymes (expressed in E. coli) are commercially available from Sigma-Aldrich, QA-Bio, AMS Biotechnology, and Merck KGaA.
|
| ● |
5X Reaction buffer for Endo-F1: 250 mM sodium phosphate buffer (pH5.5) |
| ● |
5X Reaction buffer for Endo-F2 and Endo-F3: 250 mM sodium acetate buffer (pH4.5) |
| ● |
Denaturation solution: 2% w/v sodium lauryl sulfate (SDS), 1 M β-mercaptoethanol (β-ME) |
| ● |
2X SDS-PAGE sample buffer: 0.125 M Tris-HCl buffer (pH 6.8), 10% β-ME, 4% SDS, 10% sucrose, 0.004% Bromophenol blue |
|
Instruments
 |
| ● |
Reaction incubator or water bath (100°C, 37°C) |
| ● |
|
| ● |
Microcon Ultracel YM-10 (Merck Millipore, Billerica, MA) |
|
| Methods |
|
1. |
Release of oligosaccharides from glycoproteins by using Endo-F1, -F2, or -F3 |
| 1) |
Transfer 20 μL of glycoprotein sample (10 μg/μL), 10 μL of 5X reaction buffer, and 17.5 μL of deionized water into a microtube. |
Comment 1
|

|
| 2) |
Add 2 μL of Endo-F1, Endo-F2, or Endo-F3 solution to attain a final enzyme concentration of 0.5–10 U /mL. |
Comment 0
|

|
| 4) |
Terminate the reaction by heating at 100°C for 3 min. |
Comment 1
|

|
| 5) |
Separate the oligosaccharides from the deglycosylated protein by ultrafiltration using Microcon Ultracel YM-10 (10-kDa cut-off membrane; Millipore). |
Comment 1
|
|
|
|
2. |
SDS-PAGE analysis of glycoproteins deglycosylated with Endo-F1, -F2, or -F3 |
| 1) |
Transfer 20 μL of glycoprotein sample (10 μg/μL), 10 μL of 5X reaction buffer, 2.5 μL of denaturation solution, and 15.5 μL of deionized water into a microtube. |
Comment 1
|

|
| 3) |
Add 2 μL of Endo-F1, Endo-F2, or Endo-F3 solution to attain a final enzyme concentration of 0.5–10 U /mL. |
Comment 0
|

|
| 5) |
Add 50 μL of 2X SDS-PAGE sample buffer, and then heat at 100°C for 3 min. |
Comment 0
|

|
| 6) |
Load 10 μL of the sample on SDS-PAGE gel and run the electrophoresis. Perform either Coomassie blue staining or silver staining. |
Comment 0
|
|
|
| Figure & Legends |
Figure & Legends 
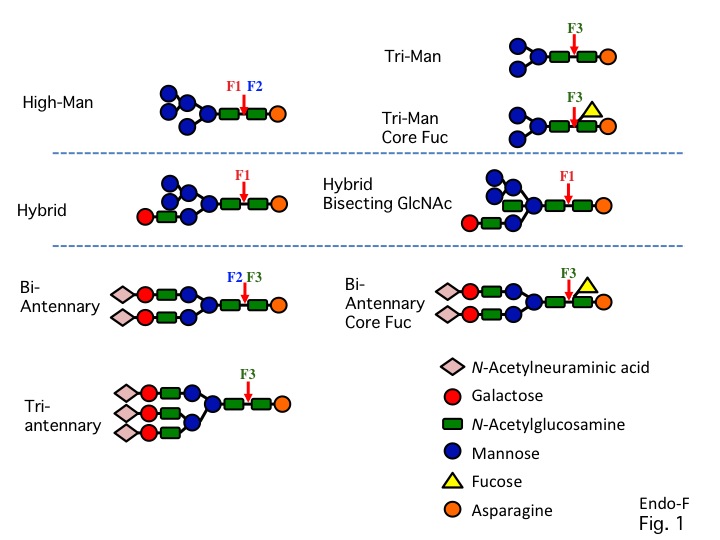
Fig. 1. Specificity of Endo-F1, -F2, and -F3. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2013-11-15 16:44:58 |
- Tarentino, A. L. and Plummer, T. H., Jr. (1994) Enzymatic deglycosylation of asparagine-linked glycans: purification, properties, and specificity of oligosaccharide-cleaving enzymes from Flavobacterium meningosepticum. Methods Enzymol. 230, 44-57 [PMID : 8139511]
- Trimble, R. B. and Tarentino, A. L. (1991) Identification of distinct endoglycosidase (endo) activities in Flavobacterium meningosepticum: endo F1, endo F2, and endo F3. Endo F1 and endo H hydrolyze only high mannose and hybrid glycans. J. Biol. Chem. 266, 1646-1651 [PMID : 1899092]
- O'Neill, R. A. (1996) Enzymatic release of oligosaccharides from glycoproteins for chromatographic and electrophoretic analysis. J. Chromatogr. A. 720, 201-215 [PMID : 8601190]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Fujita, Kiyotaka,
Yamamoto, Kenji,
(2013). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.23,4,2024 .
How to Cite this Work in Website:
Fujita, Kiyotaka,
Yamamoto, Kenji,
(2013).
Endo-β-N-acetylglucosaminidase F digestion (Endo-F).
Retrieved 23,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t73.
html source
Fujita, Kiyotaka,
Yamamoto, Kenji,
(2013).
<b>Endo-β-<em>N</em>-acetylglucosaminidase F digestion (Endo-F)</b>.
Retrieved 4 23,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t73" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t73</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|