Annexins are calcium-dependent membrane binding proteins involved in a variety of physiological processes. Membrane phospholipids, especially phosphatidylserine, are well-studied ligands for annexins. Other binding partners for annexins are particular glycans. Two methods (1. co-precipitation test and 2. solid phase system) for assaying phospholipid-binding activity of annexins are shown. |
| Category | Sugar binding proteins |
| Protocol Name | Binding of Annexin to Phopholipids |
Authors
 |
Nakano, Yukiko
Graduate School of Humanities and Sciences, Ochanomizu University
Kojima-Aikawa, Kyoko
*
Graduate School of Humanities and Sciences, Ochanomizu University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
[1. Co-precipitation test] Phospholipid: phosphatidylcholine, PC (Sigma-Aldrich, St. Louis, MO) |
| ● |
[1. Co-precipitation test] Phospholipid: phophatidylserine, PS (Sigma-Aldrich) |
| ● |
[2. Microtiter plate assay] Phophatidylserine, PS (Sigma-Aldrich) |
| ● |
[2. Microtiter plate assay] Bovine serum albumin, BSA (Wako Pure Chemical Industries, Ltd., Osaka, Japan) |
| ● |
[2. Microtiter plate assay] o-phenylenediamine, OPD (Wako Pure Chemical Industries, Ltd.) |
| ● |
[2. Microtiter plate assay] Primary antibody: rabbit anti-GST polyclonal antibody (prepared in our Lab) |
| ● |
[2. Microtiter plate assay] Secondary antibody: HRP-conjugated anti-rabbit IgG antibody (KPL, Kirkegaard & Perry Laboratories Inc., Gaithersburg, MD) |
| ● |
[2. Microtiter plate assay] Plastic micro titer plate (Immuron 1B, Thermo Fosher Scientific Inc., Waltham, MA) |
|
Instruments
 |
| ● |
[1. Co-precipitation test] Sonicator (Bransonic, Branson Ultrasonics Corporation, Danbury, CT) |
| ● |
[1. Co-precipitation test] Slab gel electrophoresis apparatus (ATTO Corporation, Tokyo, Japan) |
| ● |
[1. Co-precipitation test] Micro titer plate reader |
| ● |
[1. Co-precipitation test] Block heater |
|
| Methods |
|
1. |
|
| 1) |
Suspend phospholipids (PC:PS = 1:1, 250 μg/mL) in TBS (Tris-buffered saline; 10 mM Tris-HCl pH 7.4, 150 mM NaCl) with a sonicator for 15 min to prepare phospholipid vesicles. |
Comment 0
|

|
| 2) |
Take 20 μL of the above phospholipid solution and add 20 μL of annexin (100 μg/mL) in TBS containing 2 mM CaCl2. |
Comment 0
|

|
| 3) |
Incubate the mixture at 37°C for 30 min with occasional mixing. |
Comment 0
|

|
| 4) |
Centrifuge the mixture at 15,000 × g for 10 min, and then remove the supernatant. |
Comment 0
|

|
| 5) |
Remove unbound annexin by washing twice. Add 40 μL of TBS containing 1 mM CaCl2 to the pellet, mix gently, and centrufuge at 15,000 × g for 10 min. |
Comment 0
|

|
| 6) |
The pellet was subjected to SDS-gel electrophoresis by Laemmli’s method to detect annexin bound to phospholipids. |
Comment 0
|
|
|
|
2. |
Microtiter plate assay (semi-quantitative ELISA method using recombinant GST (glutathione S-transferase) -fused annexin and anti-GST polyclonal antibody) |
| 1) |
Add 50 μL of methanol solution of PS (50–5,000 ng) to each well of a micro titer plate. |
Comment 0
|

|
| 2) |
Bind PS to the bottom of the wells by warming the plate at 37°C on a block heater and evaporating off methanol. All other procedures were performed at room temperature. |
Comment 0
|

|
| 3) |
Wash the plate three times with TBS (Tris-buffered saline; 10mM Tris-HCl, pH7.4, 150mM NaCl). The solutions and washes can be removed by flicking the plate over a suitable waste container or a sink. |
Comment 0
|

|
| 4) |
Add 300 μL of 5% BSA/TBS and incubate for 1 h. |
Comment 0
|

|
| 5) |
Add 100 μL GST-annexin (1 μg/mL) in TBS-Ca (5mM CaCl2/TBS) containing 2.5% BSA to each well and incubate for 1h. |
Comment 1
|

|
| 6) |
Wash the plate three times with TBS-Ca. |
Comment 0
|

|
| 7) |
Add 100 μL of the primary antibody (anti-GST polyclonal antibody) at a suitable dilution with 2.5% BSA/TBS-Ca containing to each well and incubate for 1 h. |
Comment 1
|

|
| 8) |
Wash the plate three times with TBS-Ca. |
Comment 0
|

|
| 9) |
Add 300 μL of 5% BSA/TBS-Ca and incubate for 20 min. |
Comment 0
|

|
| 10) |
Add 100 μL of the secondary antibody (HRP-conjugated anti-rabbit IgG antibody) at a suitable dilution with 2.5% BSA/TBS-Ca and incubate for 30 min. |
Comment 1
|

|
| 11) |
Wash the plate three times with TBS-Ca. |
Comment 0
|

|
| 12) |
Add 150 μL of OPD solution [0.04% OPD, 0.08% H2O2/ 100mM Citrate-Phosphate buffer (pH5.0)] to each well. Incubate for 5–10 min. Positives appear orange color. |
Comment 1
|

|
| 13) |
Add 50μL of 4M H2SO4 to each well to stop enzyme reaction. |
Comment 0
|

|
|
|
| Figure & Legends |
Figure & Legends 
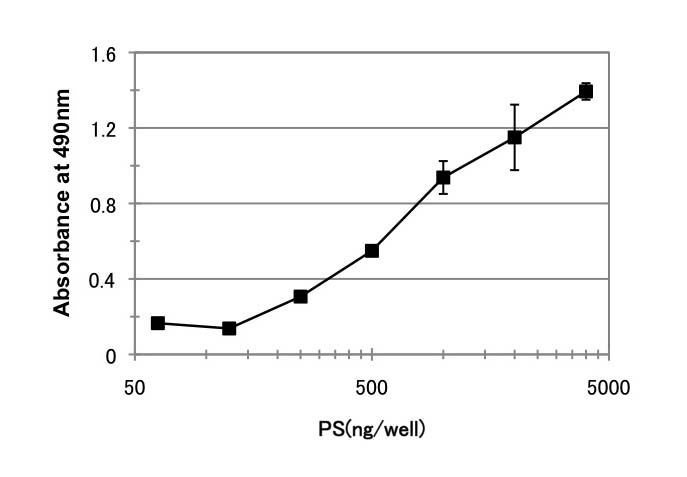
Fig. 1. Binding of GST-human annexin 5 to phosphatidylserine
GST-human annexin 5 binding to phosphatidylserine was examined. Various amounts of phosphatidylserine were immobilized on a plastic plate and allowed to react with GST-human annexin5. Each point is the mean ± SD of three determinations. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-12-18 13:47:53 |
- Kojima, K., Ogawa, H.K., Seno, N., Yamamoto, K., Irimura, T., Osawa, T., and Matsumoto, I. (1992) Carbohydrate-binding proteins in bovine kidney have consensus amino acid sequences of annexin family proteins. J Biol Chem. 267, 20536–20539 [PMID : 1400371]
- Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227, 680–685 [PMID : 5432063]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Nakano, Yukiko,
Kojima-Aikawa, Kyoko,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.19,4,2024 .
How to Cite this Work in Website:
Nakano, Yukiko,
Kojima-Aikawa, Kyoko,
(2014).
Binding of Annexin to Phopholipids.
Retrieved 19,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t7.
html source
Nakano, Yukiko,
Kojima-Aikawa, Kyoko,
(2014).
<b>Binding of Annexin to Phopholipids</b>.
Retrieved 4 19,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t7" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t7</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|