Since molecular cloning of glycosyltransferase enzymes was successfully achieved, it has become possible to directly observe the reaction, substrate specificity, enzyme kinetics and interaction with other molecules (Taniguchi N. et al. 2001). Here, methods for measurement of glycosyltransferase activity were described by selecting representative enzymes responsible for the synthesis of glycosphingolipids. |
| Category | Glycosyltransferases & related proteins |
| Protocol Name | Enzyme assay of glycolipid glycosyltransferases ~GD3 synthase (α2,8-sialyltransferase) |
Authors
 |
Furukawa, Koichi
*
Department of Biochemistry II, Nagoya University Graduate School of Medicine
Furukawa, Keiko
Deparment of Biomedical Sciences, College of Life and Health Sciences, Chubu University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Sodium cacodylate (Wako Pure Chemical Industries, Ltd., Osaka, Japan) |
| ● |
CMP-NeuAc (Sigma-Aldrich, St. Louis, MO) |
| ● |
GM3 as an acceptor (Sigma-Aldrich, St. Louis, MO) |
| ● |
[14C]-CMP-NeuAc (NEN/PerkinElmer, Waltham, MA) |
| ● |
Extracts as an enzyme source |
| ● |
Triton CF54 (Sigma-Aldrich) |
|
Instruments
 |
| ● |
|
| ● |
|
| ● |
|
| ● |
|
| ● |
|
| ● |
Ultracentrifuge and swing type bukette |
| ● |
SepPak C18 cartridge (Waters Corp., Milford, MA) |
|
| Methods |
|
1. |
Preparation of membrane fraction |
| 1) |
N2 cavitation of cell pellets at 400 psi on ice for 30 min. |
Comment 1
|

|
| 2) |
Centrifuge at 1,000 rpm for 10 min at 4°C. |
Comment 0
|

|
| 3) |
Ultracentrifuge the supernatant at 34 K (Beckman, SW 55Ti) for 1 h at 4°C. |
Comment 0
|

|
| 4) |
Resuspend the pellets in 0.1 M cacodylate buffer, pH 6.0. |
Comment 0
|
|
|
|
2. |
|
| 1) |
Evaporate the following items in a glass tube.
- CMP-NeuNc (500 μM)
- GM3 (325 μM)
- [14C]-CMP-NeuNc (9.2Å-103 dpm)
|
Comment 0
|

|
| 2) |
Dissolve in 0.05 M cacodylate buffer (adjust to make final volume 50 μL). |
Comment 0
|

|
| 3) |
Add 5 mM MnCl2, 0.3% Triton CF54 in cacodylate buffer. |
Comment 0
|

|
| 6) |
Incubate at 37°C for 2-3 h with shaking. |
Comment 0
|

|
| 7) |
Add DW 1 mL to stop the reaction. |
Comment 0
|

|
| 8) |
Separate the products by SepPak C18 column. |
Comment 0
|

|
| 10) |
Count radioactivity of 1/5 of the products by scintillation counter. |
Comment 0
|

|
| 11) |
Analyze 4/5 of the products in TLC and autoradiography. |
Comment 1
|
|
|
| Notes | This protocol was reported in the previous article (Furukawa K. et al. 2009).
Enzyme reaction of GD3 synthase was shown in Fig. 1. |
| Figure & Legends |
Figure & Legends 
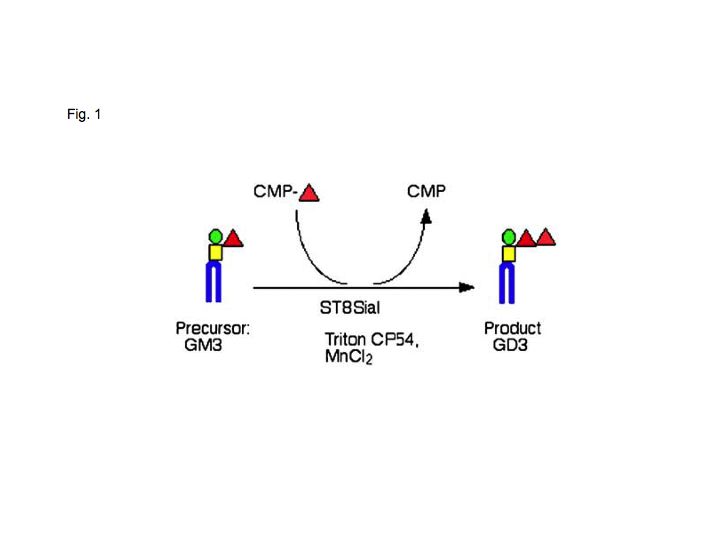
Fig. 1. Enzyme reaction of GD3 synthase (ST8SiaI)
A precursor structure GM3, sugar nucleotide CMP-NeuAc (donor substrate) and enzyme source are essential. Detergent (Triton CF54) and MnCl2 increase enzyme activity.
This figure was originally published in Glycoconj J. 26(8), 2009, 987-98, "Glycoconjugate glycosyltransferases", Furukawa K. et al. Fig.1; With kind permission of Springer Science and Business Media.
|
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2013-12-24 16:48:29 |
- Taniguchi, N., Honke, K., and Fukuda, M. (2001) Handbook of Glycosyltransferases And Related Genes. Springer-Verlag, Tokyo
- Ruan, S., and Lloyd, K.O. (1992) Glycosylation pathways in the biosynthesis of gangliosides in melanoma and neuroblastoma cells: relative glycosyltransferase levels determine ganglioside patterns. Cancer Res. 52, 5725–5731 [PMID : 1394196]
- Furukawa, K., Tsuchida, A., Okajima, T., and Furukawa, K. (2009) Glycoconjugate glycosyl-transferases. Glycoconj. J. 26, 987-998 [PMID : 18683045]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Furukawa, Koichi,
Furukawa, Keiko,
(2013). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.26,4,2024 .
How to Cite this Work in Website:
Furukawa, Koichi,
Furukawa, Keiko,
(2013).
Enzyme assay of glycolipid glycosyltransferases ~GD3 synthase (α2,8-sialyltransferase).
Retrieved 26,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t55.
html source
Furukawa, Koichi,
Furukawa, Keiko,
(2013).
<b>Enzyme assay of glycolipid glycosyltransferases ~GD3 synthase (α2,8-sialyltransferase)</b>.
Retrieved 4 26,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t55" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t55</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|