CMP-Neu5Ac hydroxylase is an enzyme responsible for the conversion of CMP-Neu5Ac to CMP-Neu5Gc, which is the key step for the expression of Neu5Gc-containing glycoconjugates in animal tissues (Naito et al., 2007). The conversion is mediated by an electron transport system consisting of cytochrome b5 reductase(s), cytochrome b5 and CMP-Neu5Ac hydroxylase using either NADH or NADPH as an electron donor. The enzyme assay method is based on the separation of CMP-Neu5Ac and CMP-Neu5Gc on a C-18 reversed-phase HPLC column (Kozutsumi et al., 1990) |
| Category | Glycosyltransferases & related proteins |
| Protocol Name | Enzyme assay of CMP-Neu5Ac hydroxylase |
Authors
 |
Takematsu, Hiromu
Laboratory of Biochemistry, Human Health Sciences, Graduate School of Medicine, Kyoto University
|
| KeyWords |
|
Reagents
 |
|
Instruments
 |
|
| Methods |
|
1. |
|
| 1) |
40 μL of Sample or the sample buffer (10 mM Tris-HC1 buffer, pH 7.5 containing 0.1 mM DTT). |
Comment 1
|

|
| 2) |
Start the reaction by adding 10 μL of the reaction mixture (10 mM Tris-HC1 buffer, pH 7.5 containing 34 nmol of NADH (or NADPH), 2 nmol of CMP-Neu5Ac and 5 mM DTT) and incubating at 37°C. |
Comment 0
|

|
| 3) |
Stop the reaction by mixing with 0.3 mL of ice-cold ethanol. |
Comment 0
|

|
| 4) |
Incubate the reaction tubes on ice for 15 min. |
Comment 0
|

|
| 5) |
Centrifuge the reaction tubes at 12,000 × g for 5 min in a refrigerated centrifuge. |
Comment 0
|
|
|
|
2. |
|
| 1) |
Analyze aliquots of the supernatant directly by HPLC on a reversed-phase column (4.6 mm i.d. × 250 mm, TSK-gel ODS-80TM; Tosoh Corp., Tokyo, Japan) using the elution buffer (50 mM NH4H2PO4) at the flow rate of 0.5 mL/min. |
Comment 0
|

|
| 2) |
Monitor CMP-Neu5Ac and CMP-Neu5Gc at 271 nm (Fig. 1) |
Comment 0
|

|
|
|
| Figure & Legends |
Figure & Legends

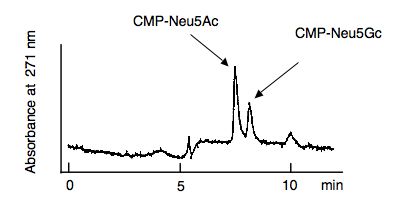
Fig. 1. A typical example of the elution profile of the reaction product. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-11-26 15:52:02 |
- Kozutsumi, Y., Kawano, T., Kawasaki, H., Suzuki, K., Yamakawa, T., and Suzuki, A. (1991). Reconstitution of CMP-N-acetylneuraminic acid hydroxylation activity using a mouse liver cytosol fraction and soluble cytochrome b5 purified from horse erythrocytes. J Biochem (Tokyo) 110, 429–435 [PMID : 1769971]
- Kozutsumi, Y., Kawano, T., Yamakawa, T., and Suzuki, A. (1990). Participation of cytochrome b5 in CMP-N-acetylneuraminic acid hydroxylation in mouse liver cytosol. J Biochem (Tokyo) 108, 704–706 [PMID : 1964451]
- Naito, Y., Takematsu, H., Koyama, S., Miyake, S., Yamamoto, H., Fujinawa, R., Sugai, M., Okuno, Y., Tsujimoto, G., Yamaji, T., et al. (2007). Germinal center marker GL7 probes activation-dependent repression of N-glycolylneuraminic acid, a sialic acid species involved in the negative modulation of B-cell activation. Mol Cell Biol 27, 3008–3022 [PMID : 17296732]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Takematsu, Hiromu,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.18,4,2024 .
How to Cite this Work in Website:
Takematsu, Hiromu,
(2014).
Enzyme assay of CMP-Neu5Ac hydroxylase.
Retrieved 18,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t231.
html source
Takematsu, Hiromu,
(2014).
<b>Enzyme assay of CMP-Neu5Ac hydroxylase</b>.
Retrieved 4 18,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t231" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t231</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|