Glycosylation mainly occurs in the endoplasmic reticulum and the Golgi complex, where it involves the sequential synthesis of glycans by glycosylation-related proteins, such as glycosyltransferases, glycosidases and nucleotide-sugar transporters in a step-wise manner. The endoplasmic reticulum and Golgi cisternae are separate compartments that specialize in different glycosylation reactions. Therefore, it is crucial for understanding in vivo glycosylation to determine in which compartments glycosylation-related proteins are localized. The current protocol provides a way to identify in which compartment a glycosylation-related protein of interest is localized in Drosophila tissues. |
| Category | Glycosyltransferases & related proteins |
| Protocol Name | Immunological analysis of glycosylation-related proteins in Drosophila tissues |
Authors
 |
Yamamoto-Hino, Miki
Department of Physiology, School of Medicine, Keio University
Okano, Hideyuki
Department of Physiology, School of Medicine, Keio University
Goto, Satoshi
*
Department of Life Science, Rikkyo University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
PBS: phosphate buffered saline |
| ● |
Fixative: 4% paraformaldehyde in PBS |
| ● |
Washing solution (PBST): 0.1% TritonX-100 or Tween20 in PBS |
| ● |
Blocking solution: 0.5% BSA (Bovine serum albumin) in PBST |
| ● |
Antibodies: anti-GM130, anti-dGLG1, anti-Syx16, anti-rabbit IgG, anti-rat IgG |
| ● |
Mounting medium: vectashield (Vector Laboratories, Inc., Burlingame, CA) |
|
| Methods |
|
1. |
Fixation of Drosophila tissues |
| 1) |
Dissect Drosophila larvae or adults in PBS. |
Comment 0
|

|
| 2) |
Fix tissues in fixative at room temperature for 20–40 min. |
Comment 0
|

|
| 3) |
Wash tissues in washing solution for 10 min three times. |
Comment 0
|
|
|
|
2. |
Immunostaining of Drosophila tissues |
| 1) |
Block tissues with blocking solution at room temperature for 30 min. |
Comment 0
|

|
| 2) |
Incubate tissue with the appropriate first antibody at 4°C for more than 8 h. |
Comment 0
|

|
| 3) |
Wash tissues in washing solution for 10 min three times. |
Comment 0
|

|
| 4) |
Incubate tissue with the appropriate secondary antibody at 4°C or room temperature for 2 h or for more than 4 h, respectively. |
Comment 0
|
|
|
|
3. |
Observation of Drosophila tissues |
| 1) |
Mount stained tissue with mounting medium. |
Comment 0
|

|
| 2) |
Observe of tissue through a laser scanning confocal microscopy. |
Comment 0
|
|
|
| Discussion | In Drosophila cells, anti-GM130, anti-dGLG1 and anti-Syx16 clearly stained cis-, medial- and trans-cisternae, respectively (Yano et al., 2005; Yamamoto-Hino et al., 2012). Co-staining using antibodies against these cisternal markers and proteins or tagged proteins of interest reveals in which cisternae the proteins of interest are localized. |
| Figure & Legends |
Figure & Legends

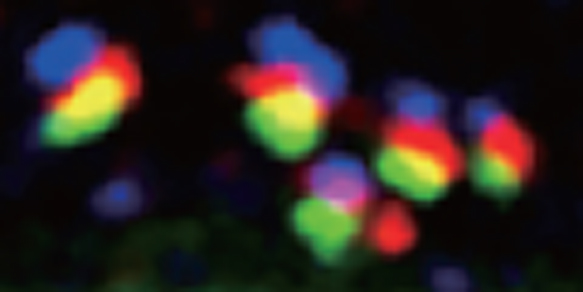
Fig. 1. Stacks of Golgi cisternae in Drosophila disc cells.
GM130 (blue), dGLG1 (red) and PNA-reactive glycans (green) are localized to cis-, medial- and trans-cisternae of the Golgi complex in Drosophila disc cells.
This figure was originally published in Proc. Natl. Acad. Sci. Yano H, Goto S et al. "Distinct functional units of the Golgi complex in Drosophila cells" 2005, 102(38):13467-72 ©the National Academy of Sciences. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-05-27 13:16:47 |
- Yamamoto-Hino, M., Abe, M., Shibano, T., Setoguchi, Y., Awano,W., Ueda.R., Okano, H. and Goto, S. (2012) Cisterna-specific localization of glycosylation-related proteins to the Golgi apparatus. Cell Struct. Funct. 37, 55–63. [PMID : 22251795]
- Yano, H., Yamamoto-Hino, M., Abe, M., Kuwahara, R., Haraguchi, S., Kusaka, I., Awano, W., Kinoshita-Toyoda, A., Toyoda, H. and Goto, S. (2005) Distinct functional units of the Golgi complex in Drosophila cells. Proc. Natl. Acad. Sci. USA. 102, 13467–13472. [PMID : 16174741]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Yamamoto-Hino, Miki,
Okano, Hideyuki,
Goto, Satoshi,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.19,4,2024 .
How to Cite this Work in Website:
Yamamoto-Hino, Miki,
Okano, Hideyuki,
Goto, Satoshi,
(2014).
Immunological analysis of glycosylation-related proteins in Drosophila tissues.
Retrieved 19,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t184.
html source
Yamamoto-Hino, Miki,
Okano, Hideyuki,
Goto, Satoshi,
(2014).
<b>Immunological analysis of glycosylation-related proteins in <em>Drosophila </em>tissues</b>.
Retrieved 4 19,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t184" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t184</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|