The structural homogeneity and common occurrence in vertebrates make the production of antibody against hyaluronan (HA) impossible. Fortunately, there are a number of naturally occurring HA-binding proteins (HABP) that exhibit high specificity and affinity to HA and, therefore, can be used in immunoassay as a substitute of the antibody. The firstly and most widely used protein is the HA-binding regions (HABR) of cartilage proteoglycan (aggrecan) and link protein introduced by Tengblad in 1979 1). Using HABPs, various quantification assay methods for HA have been developed and have given satisfying results 2). According to the working principle, these systems can be divided into three categories: competitive RIA/ELISA3), sandwich ELISA 4) and latex agglutination 5). The first two are traditional assay systems used by researchers in the world wide, while the third is recently developed and mainly used in clinical testing. Here we provide example protocols of the first and second categories. It is noteworthy that the sandwich ELISA requires at least two HABP-binding sites in the target HA, while the competitive ELISA requires only one site. The results obtained by sandwich ELISA may be less than the true level if the sample includes HA smaller than 10kDa. In this case, the competitive ELISA is recommended. |
| Category | Glycosaminoglycans |
| Protocol Name | Application of anti-GAG antibody and biotinylated hyaluronan binding protein(bHABP) [2] ~ ELISA for hyaluronan quantification |
Authors
 |
Zhuo, Lisheng
*
Research complex for Medicine Fontiers, Aichi Medical University
Kimata, Koji
Research Complex for the Medicine Frontiers, Aichi Medical University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Hyaluronan binding protein (HABP) and biotinylated HABP (b-HABP) (Cat#400762 & #400763: Seikagaku Corp., Tokyo, Japan ) |
| ● |
Streptavidin conjugated horse radish peroxidase(HRP) (Cat#016-030-084: Jackson ImmunoResearch Laboratories Inc., West Grove, PA) |
| ● |
Sureblue® TMB microwell peroxidase substrate (KPL #52-00-02) |
| ● |
N-hydroxysulfosuccinimide Na (SulfoNHS, CAS#106627-54-7) |
| ● |
1-ethyl-3-(3-dimethylaminopropyl)carbodiimide, hydrochloride salt (EDC, CAS#25952-53-8) |
| ● |
HA standards (10-640 ng/mL) |
|
Instruments
 |
| ● |
Nunc F96 Maxisorp Immunoplate (Cat#442404) |
| ● |
Sumitomo Becklite ELISA plate type A (Cat#MS-8696F) |
| ● |
|
|
| Methods |
|
1. |
|
| 1) |
Nunc Maxisorp Immunoplate is coated with 50 μL/well of HABP solution (0.2 μg/mL in 0.1M NaHCO3, pH 9.3) overnight at 4°C, followed by washing with PBS containing 0.1% Tween 20 (PBST) (200 μL/well) twice. |
Comment 0
|

|
| 2) |
The wells are blocked with PBST containing 2% BSA (200 μL/well) at 37°C for 1 h and then washed as above. |
Comment 0
|

|
| 3) |
HA standards and samples are added (50 μL/well). The plate is then incubated at 37°C for 1 h, followed by washing with PBST (200 μL/well) three times. |
Comment 0
|

|
| 4) |
b-HABP solution (0.3 μg/mL in PBST) is added (50 μL/well). The plate is then incubated and washed as above. |
Comment 0
|

|
| 5) |
Streptavidin-HRP (1:3000 diluted in PBST) is added (50 μL/well). The plate is then incubated and washed as above. |
Comment 0
|

|
| 6) |
TMB peroxidase substrate is added (50 μL/well) to develop a blue color. |
Comment 0
|

|
| 7) |
When the color density reaches a satisfying level,1M HCl (50 μL/well) is added immediately to stop the reaction. The color changes from blue to yellow. |
Comment 0
|

|
| 8) |
Absorbance at 450 nm is measured on a microplate reader. |
Comment 0
|
|
|
|
2. |
|
| 1) |
HA is immobilized on Sumitomo Becklite type A ELISA plate by mixing 25 μL of SulfoNHS (18.4 μg/mL), 25 μL of HA (0.5 μg/mL) and 50 μL of EDC (6.15 μg/mL) in each well, followed by incubating at room temperature for 2 h and then overnight at 4°C. |
Comment 0
|

|
| 2) |
The wells are washed with 2M NaCl (100 μL/well) and then distilled water (200 μL/well) three times each. |
Comment 0
|

|
| 3) |
The wells are blocked with PBST containing 2% BSA (200 μL/well) at 37°C for 1 h, followed by washing with PBST (200 μL/well) three times. |
Comment 0
|

|
| 4) |
Twenty-five μL of HA standards or samples and 25 μL of b-HABP (0.5 μg/mL in PBST) are added to each well. The plate is then incubated and washed as above. |
Comment 0
|

|
| 5) |
Streptavidin-HRP (1:3000 diluted in PBST) is added (50 μL/well). The plate is then incubated and washed as above. |
Comment 0
|

|
| 6) |
TMB peroxidase substrate is added (50 μL/well) to develop a blue color. |
Comment 0
|

|
| 7) |
When the color density reaches a satisfying level,1M HCl (50 μL/well) is added immediately to stop the reaction. The color changes from blue to yellow. |
Comment 0
|

|
| 8) |
Absorbance at 450 nm is measured on a microplate reader. |
Comment 0
|
|
|
| Initial amount | |
| Figure & Legends |
Figure & Legends

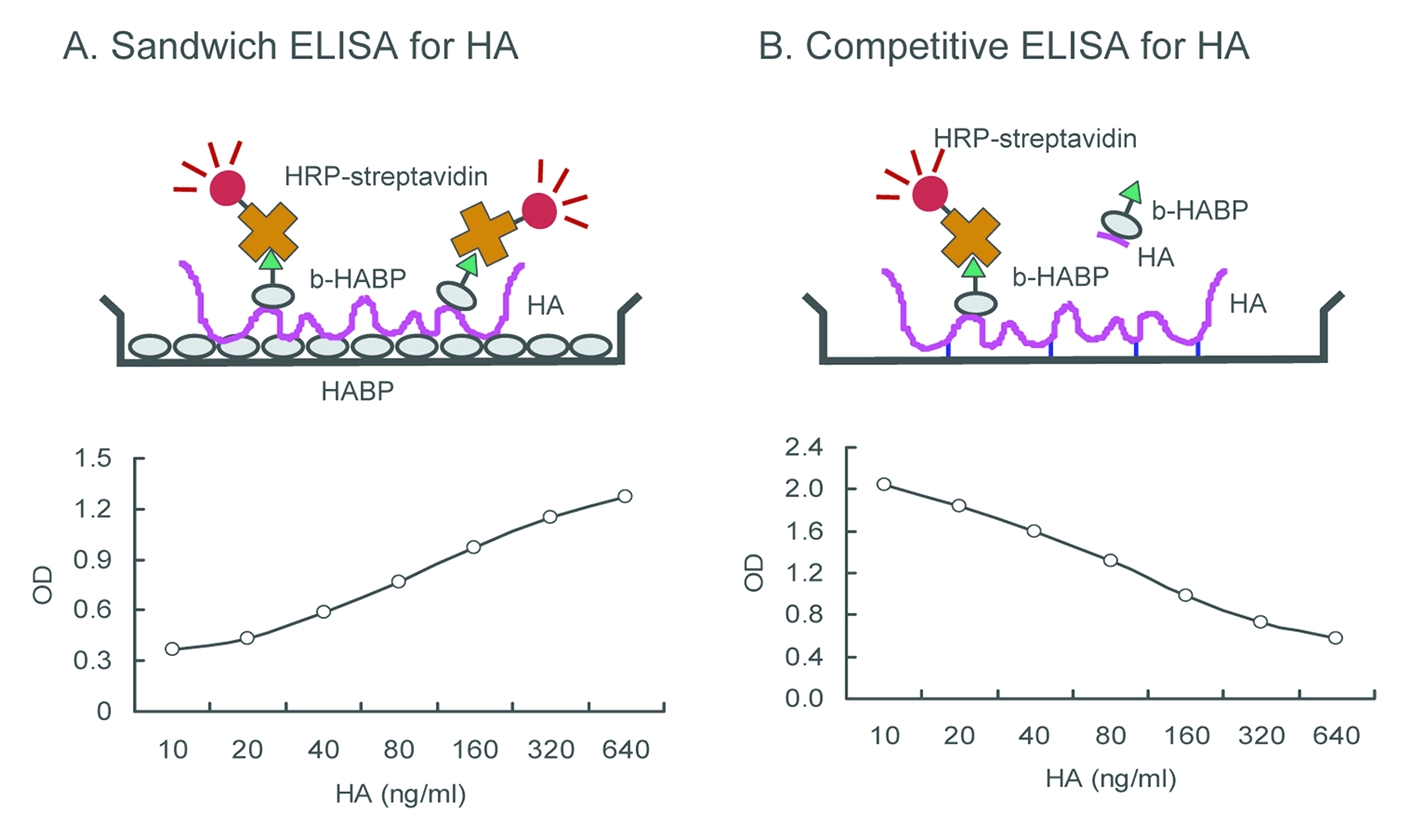
Fig. 1. Shematic representation of ELISA for HA.
(A) Sandwich ELISA. HA in the sample solutions is captured by the immobilized HABP and detected by the biotinylated HABP. The absorbance correlates positively with the level of HA. (B) Competitive ELISA. HA in the sample solutions competes with the immobilized HA for the biotinylated HABP. The absorbance correlates reversely with the level of HA. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2017-01-23 15:52:10 |
- Tengblad, A. (1979) Affinity chromatography on immobilized hyaluronate and its application to the isolation of hyaluronate binding properties from cartilage. Biochim. Biophys. Acta. 578, 281-289 [PMID : 486527]
- Lindqvist, U., Chichibu, K., Delpech, B., Goldberg, R. L., Knudson, W., Poole, A. R., and Laurent, T. C. (1992) Seven different assays of hyaluronan compared for clinical utility. Clin Chem. 38, 127-32 [PMID : 1370770]
- Tengblad, A. (1980) Quantitative analysis of hyaluronate in nanogram amounts. Biochem. J. 185,101-105 [PMID : 6445729]
- Chichibu, K., Matsuura, T., Shichijo, S., and Yokoyama, M. M. (1989) Assay of serum hyaluronic acid in clinical application. Clin Chim Acta. 181, 317-23 [PMID : 2474393]
- Kakuta, K. et al, Japan Patent P2011-090004.
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Zhuo, Lisheng,
Kimata, Koji,
(2017). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.18,4,2024 .
How to Cite this Work in Website:
Zhuo, Lisheng,
Kimata, Koji,
(2017).
Application of anti-GAG antibody and biotinylated hyaluronan binding protein(bHABP) [2] ~ ELISA for hyaluronan quantification.
Retrieved 18,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t159.
html source
Zhuo, Lisheng,
Kimata, Koji,
(2017).
<b>Application of anti-GAG antibody and biotinylated hyaluronan binding protein(bHABP) [2] ~ ELISA for hyaluronan quantification</b>.
Retrieved 4 18,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t159" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t159</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|