To elucidate the structure of the sugar moiety of O-glycoproteins, exoglycosidase digestion is useful. For example, the terminal sugar, α/β-anomer and linkage of the terminal sugar can be estimated using exoglycosidase treatment. On the other hand, a specific type of O-glycans can be removed from the peptides and/or proteins by endoglycosidase (O-glycanase or O-glycosidase). |
| Category | O-Glycans |
| Protocol Name | Glycosidase digestion of O-glycoproteins and related O-glycans |
Authors
 |
Goda M., Hatsumi
*
Department of Bioscience and Biotechnology, Graduate School of Bioresource and Bioenvironmental Sciences, Kyushu University
Ito, Makoto
Department of Bioscience and Biotechnology, Graduate School of Bioresource and Bioenvironmental Sciences, Kyushu University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Neuraminidase (Arthrobacter ureafaciens) (Sigma-Aldrich, St. Louis, MO) |
| ● |
O-glycosidase (endo-α-N-acetylgalactosaminidase) (Streptococcus pneumoniae) (Sigma-Aldrich) |
|
Instruments
 |
| ● |
Speed Vac Concentrator (Thermo Fisher Scientific Inc., Waltham, MA) |
|
| Methods |
|
1. |
Removal of sialic acids from fetuin O-glycans by Arthorobacter sialidase. |
| 1) |
Dissolve 30 μg of O-glycans (eg. Sialyl-T antigen) in 100 μL of 50 mM sodium acetate buffer, pH 5.0, with 50 mU of Arthrobacter sialidase. |
Comment 0
|

|
| 2) |
Incubate the mixture at 37°C for 16 h. |
Comment 0
|

|
| 3) |
Terminate the reaction to keep the sample in boiling water for 3 min. |
Comment 0
|

|
| 5) |
Centrifuge at 15,000 rpm for 10 min. |
Comment 0
|

|
| 6) |
Dry the supernatant with a Speed Vac Concentrator. |
Comment 0
|
|
|
|
2. |
Removal of a core I disaccharide (Galβ1-3GalNAc) from an O-glycoprotein by endo-α-N-acetylgalactosaminidase. |
| 1) |
Dissolve 300 μg of O-glycoprotein (eg. asialofetuin) in 100 μL of 50 mM sodium phosphate buffer, pH 6.0, with 50 mU of O-glycosidase. |
Comment 1
|

|
| 2) |
Incubate the mixture at 37°C for 16 h. |
Comment 0
|

|
| 3) |
Terminate the reaction to keep the sample in boiling water for 3 min. |
Comment 0
|

|
| 5) |
Centrifuge at 15,000 rpm for 10 min. |
Comment 0
|

|
| 6) |
Dry the supernatant with a Speed Vac Concentrator. |
Comment 0
|
|
|
| Figure & Legends |
Figure & Legends 
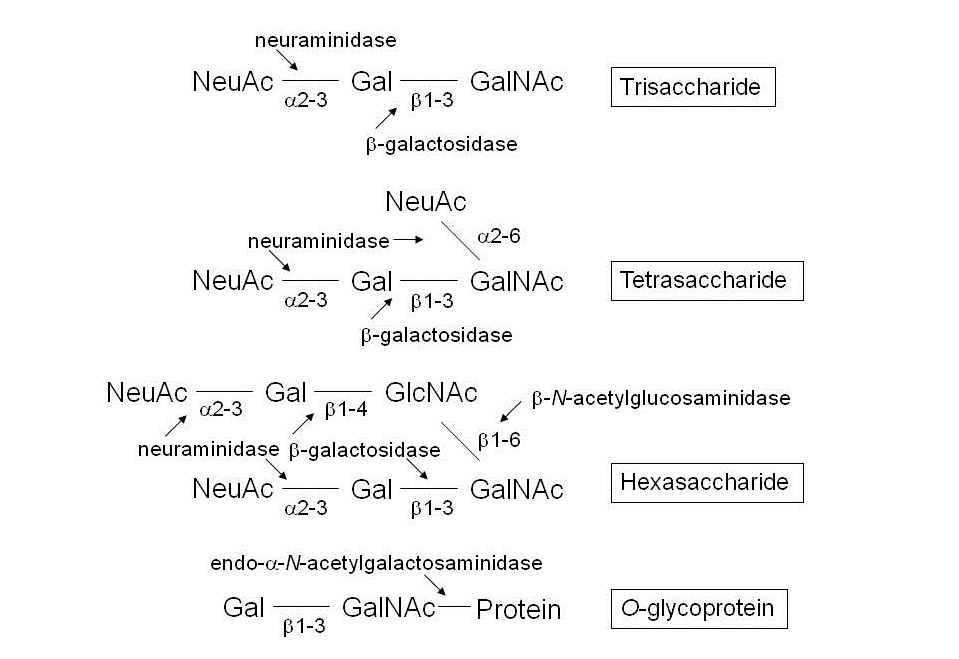
Fig. 1. Action points of glycosidases on O-glycoproteins and related O-glycans |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-04-22 10:46:42 |
- Royle L, Mattu TS, Hart E, Langridge JI, Merry AH, Murphy N, Harvey DJ, Dwek RA, and Rudd PM. (2002) An analytical and structural database provides a strategy for sequencung O-glycan from microgram quantities of glycoproteins. Anal Biochem. 304, 70-90 [PMID : 11969191]
- Edge AS, and Spiro RG. (1987) Presence of an O-glycosidically linked hexasaccharide in fetuin. J Biol Chem. 262, 16135-16141 [PMID : 2445744]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Goda M., Hatsumi,
Ito, Makoto,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.20,4,2024 .
How to Cite this Work in Website:
Goda M., Hatsumi,
Ito, Makoto,
(2014).
Glycosidase digestion of O-glycoproteins and related O-glycans.
Retrieved 20,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t14.
html source
Goda M., Hatsumi,
Ito, Makoto,
(2014).
<b>Glycosidase digestion of <em>O</em>-glycoproteins and related <em>O</em>-glycans</b>.
Retrieved 4 20,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t14" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t14</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|