Influenza virus hemagglutinin (HA) attaches to sialoglycoconjugates on the host cell surface to initiate infection. Binding specificity of influenza virus for sialooligosaccharide moieties on the cell surface is a critical factor for acquiring transmission ability to differentiate host species (Suzuki Y. et al. 1992; Suzuki T. et al. 2001; Shinya K et al. 2005; Takemae N. et al. 2009; Sriwilaijaroen N. et al. 2009). ELISA-based method for assay of receptor binding specificity of influenza viruses using sialoglycosphingolipids (gangliosides) (Suzuki Y. et al. 1992; Suzuki T. et al. 2001; Shinya K et al. 2005) as viral receptors is described in this paper. |
| Category | Sugar binding proteins |
| Protocol Name | ELISA-based assay for determination of influenza virus-host receptor binding specificity |
Authors
 |
Suzuki, Yasuo
*
Health Science Hills, College of Life and Health Sciences, Chubu University
Sriwilaijaroen, Nongluk
(1) Health Science Hills, College of Life and Health Sciences, Chubu University, (2) Faculty of Medicine, Thammasat University (Rangsit Campus)
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
|
| ● |
Blocking solution: 1 g of defatted bovine serum albumin (BSA) in 100 mL of PBS |
| ● |
Dilution buffer: 0.5 g of defatted BSA in 100 mL of PBS |
| ● |
Substrate solution: 2 mg of o-phenylenediamine (Sigma-Aldrich, St. Louis, MO) is dissolved in 10 mL of 100 mM citrate-phosphate buffer (pH5.5) before addition of 3.3 μL of 30% H2O2. This coloring solution should be mixed freshly before use. |
|
Instruments
 |
| ● |
96-well Polysorp flat-bottomed plates |
| ● |
|
| ● |
|
|
| Methods |
|
1. |
ELISA-based assay for determination of influenza virus-host receptor binding specificity |
| 1) |
Dissolve ganglioside (3–50 pmol/10 μL/well) in ethanol or another organic solvent that has no effect on a plastic plate (F96 polysorp; Nalge Nunc International) and dilute serially two-fold with ethanol. |
Comment 0
|

|
| 2) |
Evaporate ethanol at 37°C for several minutes and dry completely with warm air. |
Comment 0
|

|
| 3) |
Add 250 μL of blocking solution (PBS with 1% of defatted BSA) to each well and incubate for 2 h at 37°C. |
Comment 0
|

|
| 4) |
Wash the wells five times with PBS, add 100 μL of 25 HAU of influenza virus in PBS, and keep at 4°C overnight with gentle shaking. |
Comment 0
|

|
| 5) |
Wash unbound virus five times with cold PBS, add 100 μL of primary antibody directed to the virus diluted in 0.5% defatted BSA, and keep for 2 h at 4°C. |
Comment 0
|

|
| 6) |
Wash five times with PBS to get rid of free primary antibody, add 100 μL of HRP-conjugate secondary antibody in 0.5% defatted BSA, and keep for 2 h at 4°C. |
Comment 0
|

|
| 7) |
Wash away unbound conjugate five times with PBS, add 100 μL of coloring solution, and keep at room temperature until a yellow color develops. |
Comment 0
|

|
| 8) |
Stop the reaction by adding 50 μL of 1 N H2SO4. This acidic solution will change color from yellow to orange. |
Comment 0
|

|
| 9) |
Record the absorbance at 490 nm with reference wavelength of 630 nm. Before plotting a graph, subtract OD490 from OD630 to obtain the actual values of virus binding activity. |
Comment 0
|
|
|
| Notes | If purified, gangliosides can be coated on wells of a flat-bottomed microtiter plate and the ELISA technique can be employed for quantitative detection of virus binding activity to the gangliosides. The gangliosides and influenza virus materials are obtained from the same sources as those of the TLC test, but a smaller amount (25 HAU, influenza virus: 3–50 pmol/well, ganglioside) can be detected by ELISA. The other reagents are similar to those of TLC except for the reagents mentioned above.
Influenza viruses should be handled in the facility of Biological Safety Level 2 or 3. Therefore, handling of the viruses should be under the control of national law. |
| Figure & Legends |
Figure & Legends 
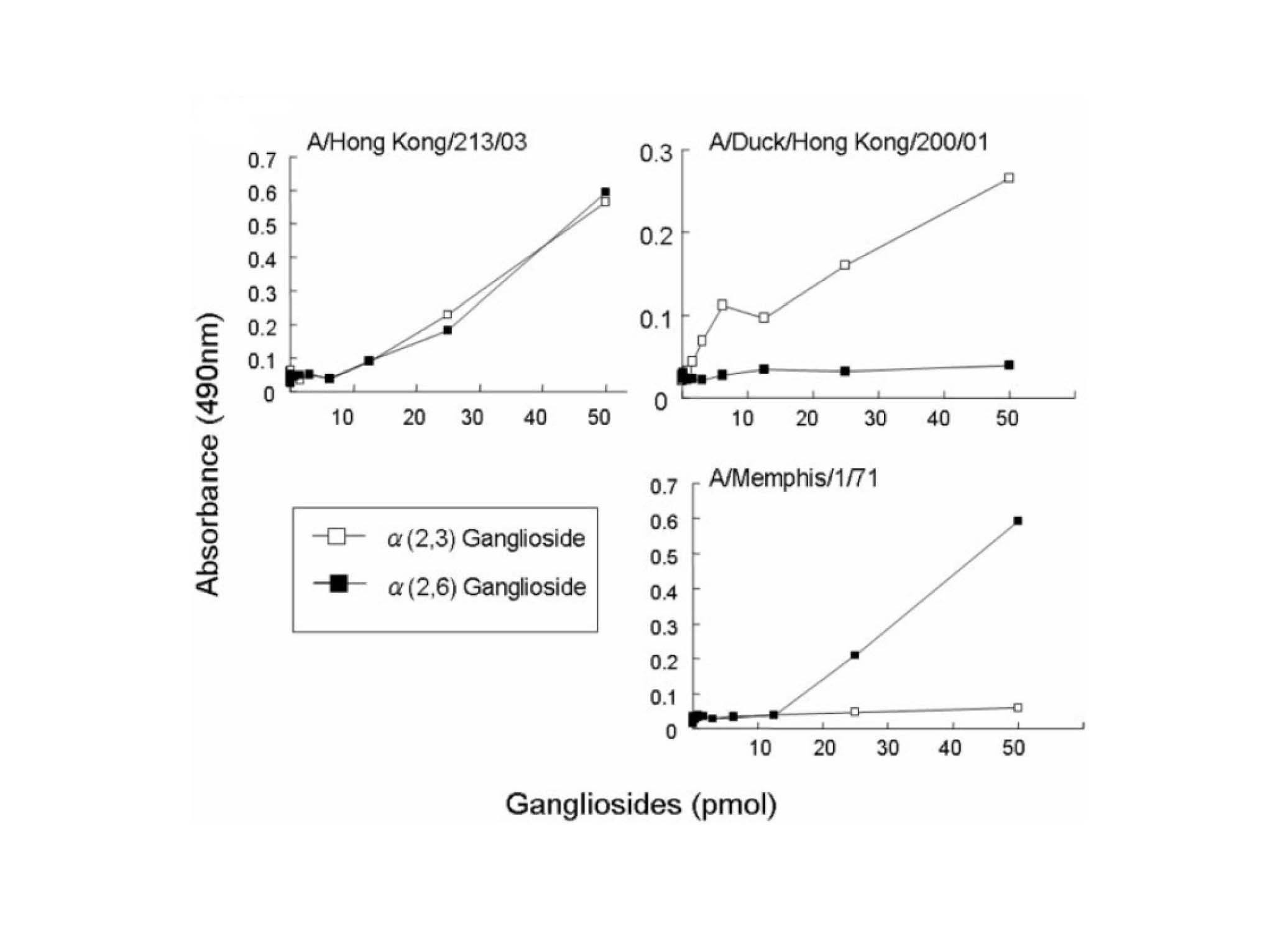
Copyright © American Society for Microbiology, Journal of Virology, 79(15), 2005, 9926–32, doi:10.1128/JVI.79.15.9926-9932.2005.
Fig. 1. Receptor binding specificity of highly pathogenic avian influenza virus, A/KongKong/213/03 (H5N1) isolated from human
The direct binding activity of viruses to sialylparagloboside (IV3(Neu5Ac)Lc4Cer, IV6(Neu5Ac)Lc4Cer) was determined as described in this protocol. A/HongKong/213/03 (H5N1) bound to both Neu5Acα2-3paragloboside (IV3(Neu5Ac)Lc4Cer), and Neu5Acα2-6paragloboside (IV6(Neu5Ac)Lc4Cer) but A/Duck/HongKong/200/01 isolated from duck bound preferentially to Neu5Acα2-3paragloboside (IV3(Neu5Ac)Lc4Cer) and A/Memphis/1/71 isolated from human bound preferentially to Neu5Acα2-6paragloboside (IV6(Neu5Ac)Lc4Cer). |
| Copyrights |
Copyright 2008. American Society for Microbiology, for a figure in [Figure & Legends].
Copyright 2011. Ritsumeikan University, JCGGDB & AIST. for the rest of the contents. |
| Date of registration:2014-10-20 16:31:08 |
- Suzuki, Y., Nakao, T., Ito, T., Watanabe, N., Toda, Y., Xu, G., Suzuki, T., Kobayashi, T., Kimura, Y., and Yamada, A. (1992) Structural determination of gangliosides that bind to influenza A, B, and C viruses by an improved binding assay: strain-specific receptor epitopes in sialo-sugar chains. Virology. 189, 121–31 [PMID : 1376537]
- Suzuki, T., Portner A., Scroggs, R.A., Uchikawa, M., Koyama, N., Matsuo, K., Suzuki, Y., and Takimoto, T. (2001) Receptor specificities of human respiroviruses. J. Virol. 75, 4604–4613 [PMID : 11312330]
- Shinya, K., Hatta, M., Yamada, S., Takada, A., Watanabe, S., Halfmann, P., Horimoto T., Neumann, G., Kim, J.H., Lim, W., Guan, Y., Peiris, M., Kiso, M., Suzuki, T., Suzuki, Y., and Kawaoka, Y. (2005) Characterization of a Human H5N1 Influenza A Virus Isolated in 2003. J. Virol. 79, 9926–9932 [PMID : 16014953]
- Takemae, N., Ruttanapumma, R., Parchariyanon, S., Yoneyama, S., Hayashi, T., Hiramatsu, H., Sriwilaijaroen, N., Uchida, Y., Kondo, S., Yagi, H., Kato, K., Suzuki, Y., and Saito, T. (2010) Alterations in receptor binding properties of swine influenza viruses of H1 subtype after isolation in embryonated chicken eggs. J. gen. Virol. doi: 10.1099/vir.0.016691-0 (Dec. 9th, 2009) [PMID : 20007353]
- Sriwilaijaroen, N., Kondo, S., Yagi, H., Wilairat, P., Hiramatsu, H., Ito, M., Ito, Y., Kato, K., Suzuki, Y. (2009) Analysis of N-glycans in embryonated chicken egg chorioallantoic and amniotic cells responsible for binding and adaptation of influenza viruses. Glycoconjugate J. 26, 433–443 [PMID : 18853253]
|
|
For those who wish to reuse the work, please contact JCGGDB management office (jcggdb-ml@aist.go.jp).
|
|