Although the first biological activity attributed to galectin-9 is its chemoattractant activity as to eosinophils, accumulating evidence has established galectin-9 as a novel type of regulator of the immune response and homeostasis. The antiproliferative effect on peripheral blood T cells (and T-cell lines) is a typical immune-modulating activity of galectin-9. Galectin-9 induces T cell apoptosis through caspase-dependent and -independent pathways. In addition, non-apoptotic mechanisms also contribute to galectin-9-induced cell death. The present protocol does not discriminate the antiproliferative mechanisms, and thus it is easy to perform and reproducible. |
| Category | Sugar binding proteins |
| Protocol Name | Assay method for the antiproliferative activity of human galectin-9 |
Authors
 |
Nishi, Nozomu
*
Division of Research Instrument and Equipment, Life Science Research Center, Kagawa University
Nakamura, Takanori
Department of Endocrinology, Faculty of Medicine, Kagawa University
*To whom correspondence should be addressed.
|
| KeyWords |
|
Reagents
 |
| ● |
Jurkat T lymphoma (lymphoblastoma) cells |
| ● |
RPMI 1640* supplemented with 10% FBS, 100 units/mL penicillin, 100 μg/mL streptomycin (RPMI 1640 medium)
* With L-glutamine and sodium bicarbonate (Ex. Sigma-Aldrich R8758) |
| ● |
Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan) |
| ● |
1.2% (w/v) sodium dodecyl sulfate (SDS) |
|
Instruments
 |
| ● |
|
| ● |
|
| ● |
|
| ● |
Inversed phase contrast microscope |
| ● |
|
|
| Methods |
|
1. |
Assay method for the antiproliferative activity of human galectin-9 |
| 1) |
Collect Jurkat cells* by centrifugation at 150 × g for 5 min using the tabletop centrifuge (*see Comment). |
Comment 1
|

|
| 2) |
Resuspend the cell pellet in fresh RPMI 1640 medium. |
Comment 0
|

|
| 3) |
Determine the cell concentration with a hemocytometer. |
Comment 0
|

|
| 4) |
Add an appropriate volume of the medium to make the final concentration 3 × 104 cells/90 μL. |
Comment 0
|

|
| 5) |
Inoculate the cell suspension into a 96-well tissue culture plate (90 μL/well) using a multi-channel pipette. |
Comment 0
|

|
| 6) |
Incubate the plate for 2 h at 37°C under a humidified 5% CO2 atmosphere (CO2 incubator). |
Comment 0
|

|
| 7) |
Add 10 μL/well of sterile test samples directly to the plate. |
Comment 1
|

|
| 8) |
Incubate the plate for 24 h* at 37°C in the CO2 incubator (*see Comment). |
Comment 1
|

|
| 9) |
Add 10 μL/well of WST-8 reagent. |
Comment 1
|

|
| 10) |
Incubate the plate for 2 h* at 37°C in the CO2 incubator (*see Comment). |
Comment 1
|

|
| 11) |
Add 10 μL/well of the 1.2% SDS solution. |
Comment 0
|

|
| 12) |
Determine the difference between the absorbance at 450 and that of 620 nm with the microplate reader. |
Comment 0
|
|
|
| Notes | This protocol can be used for other cell types including adherent cells. When an adherent cell line is used as a target for galectin-9, the incubation time after inoculation should be longer (up to 24 h) to facilitate the attachment of protease-treated cells. |
| Figure & Legends |
Figure & Legends 
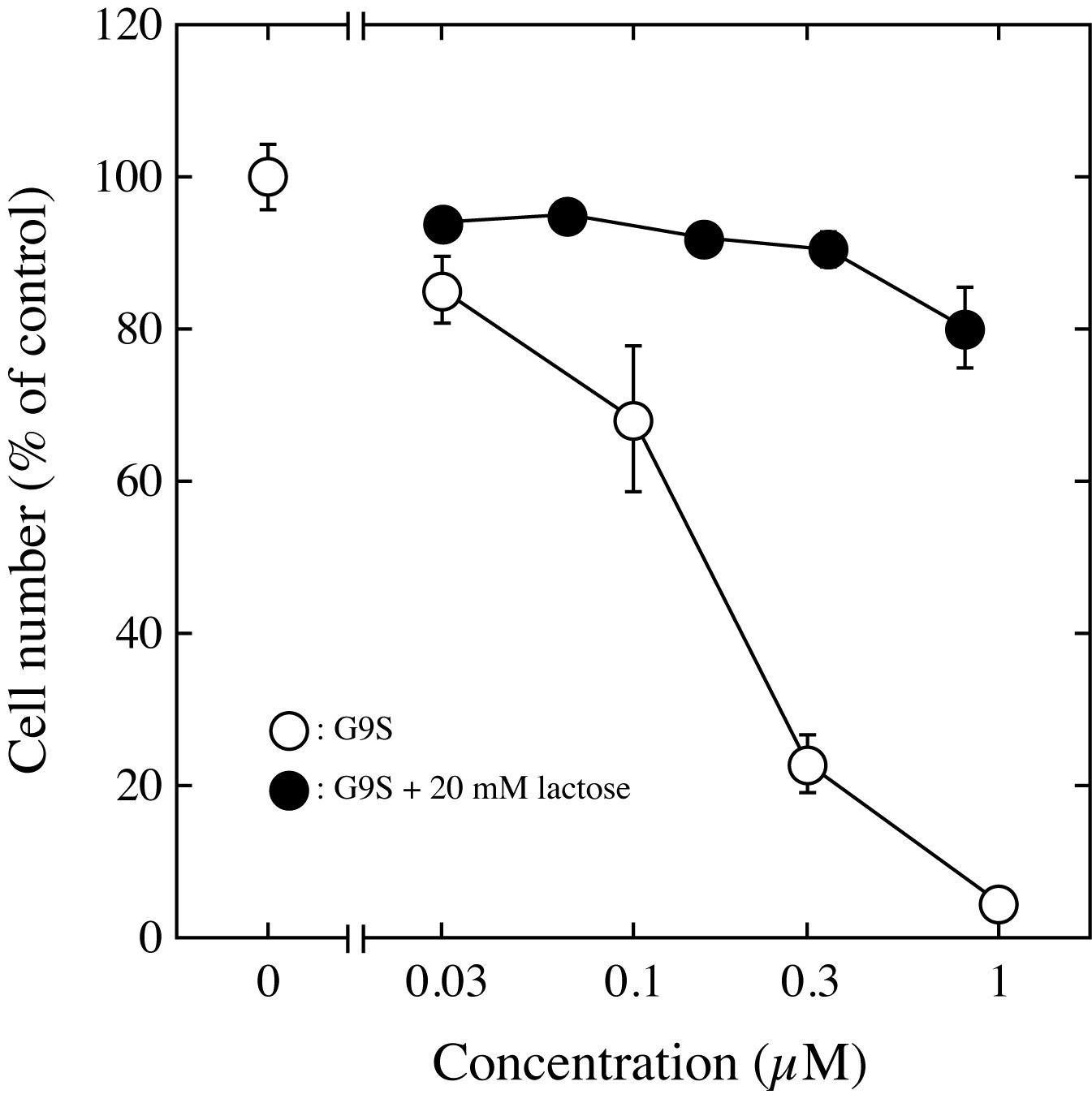
Fig. 1. The antiproliferative effect of G9S on Jurkat cells was determined by means of the WST-8 assay.
Jurkat cells were cultured in the presence of an assay sample for 24 h. After the addition of WST-8 reagent, the culture was continued for 2 h. The viable cell number was determined with a microplate reader. The viable cell number for the untreated control culture was taken as 100%. The results represent the means ± S.D. of triplicate measurements. |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2014-08-26 16:37:05 |
- Kashio, Y., Nakamura, K., Abedin, M.J., Seki, M., Nishi, N., Yoshida, N., Nakamura, T., and Hirashima, M. (2003) Galectin-9 induces apoptosis through the calcium-calpain-caspase-1 pathway. J Immunol. 170, 3631–3636 [PMID : 12646627]
- Lu, L.H., Nakagawa, R., Kashio, Y., Ito, A., Shoji, H., Nishi, N., Hirashima, M., Yamauchi, A., and Nakamura, T. (2007) Characterization of galectin-9-induced death of Jurkat T cells. J Biochem. 141, 157–172 [PMID : 17167046]
- Fukata, Y., Itoh, A., Nonaka, Y., Ogawa, T., Nakamura, T., Matsushita, O., and Nishi, N. (2014 ) Direct cytocidal effect of galectin-9 localized on collagen matrices on human immune cell lines. Biochim Biophys Acta. 1840, 1892–1901 [PMID : 24462947]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Nishi, Nozomu,
Nakamura, Takanori,
(2014). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.26,4,2024 .
How to Cite this Work in Website:
Nishi, Nozomu,
Nakamura, Takanori,
(2014).
Assay method for the antiproliferative activity of human galectin-9.
Retrieved 26,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t106.
html source
Nishi, Nozomu,
Nakamura, Takanori,
(2014).
<b>Assay method for the antiproliferative activity of human galectin-9</b>.
Retrieved 4 26,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t106" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t106</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|