Hydrazinolysis, differing from an enzymatic procedure to liberate glycans from glycoproteins, is not essentially influenced by chemical structures of glycoproteins, and this makes a basic reason for its reliability in comparison with the enzymatic procedure. Moreover, as described above, hydrazine anhydrous shows excellent performance as a solvent. |
| Category | N-Glycans |
| Protocol Name | Release of N-glycans by hydrazinolysis |
Authors
 |
Nakakita, Shin-ichi
*
Department of Functional Glycomics, Life Science Research Center, Kagawa University
Natsuka, Shunji
Department of Biology, Faculty of Science, Niigata University
*To whom correspondence should be addressed.
|
Reagents
 |
| ● |
Hydrazine Anhydrous (Tokyo Chemical Industry Co., Ltd. Tokyo, Japan) |
| ● |
Sodium Hydrogen Carbonate (Wako Pure Chemical Industries Ltd., Osaka, Japan) |
| ● |
Acetic anhydride (Wako Pure Chemical Industries Ltd.) |
|
Instruments
 |
| ● |
Speed Vac Concentrator (Thermo Fisher Scientific Inc., Waltham, MA) |
| ● |
Block incubator (Astec Co., Ltd., Fukuoka, Japan) |
| ● |
Dowex 50 W (The Dow Chemical Company, Midland, MI) |
| ● |
Rotary oil pump connected to cold trap (Yamato Scientific Co., Ltd., Tokyo, Japan) |
|
| Methods |
|
1. |
Release of N-glycan by hydrazinolysis |
| 1) |
Sample (glycoprotein or tissue) is lyophilized. |
Comment 0
|

|
| 2) |
Two milligram of powder is put on a bottom of the screw seal tube. |
Comment 0
|

|
| 3) |
Add 200 μL of the hydrazine anhydrous in screw seal tube. |
Comment 1
|

|
| 4) |
Mix hydrazine anhydrous and powder. |
Comment 0
|

|
| 6) |
Remove hydrazine anhydrous using a rotary pump connected with cold glass trap in vacuo. |
Comment 0
|

|
| 7) |
Add 0.2 mL saturated sodium bicarbonate solution and 0.016 mL acetic anhydride at 0°C for 5 min. |
Comment 0
|

|
| 8) |
Add 0.2 mL saturated sodium bicarbonate solution and 0.016 mL acetic anhydride at 0°C for 30 min. |
Comment 0
|

|
| 9) |
Add 1 g of Dowex 50 W-X2 (H+ form). |
Comment 0
|

|
| 10) |
Pour into a small glass column. |
Comment 0
|

|
| 11) |
Wash with 5 column volumes of water. |
Comment 0
|

|
| 12) |
Combine the eluate and the washings, concentrated, and lyophilized. |
Comment 0
|
|
|
| Figure & Legends |
Figure & Legends 
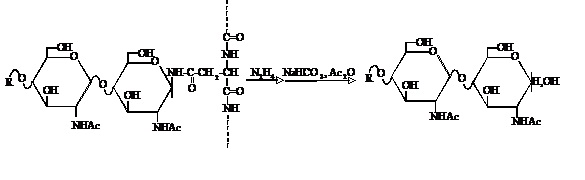
Fig. 1. Chemical liberation of N-linked oligosaccharides from glycoproteins by hydrazinolysis- N-acetylation
This figure was originally published in "Mirai wo Hiraku Tousa Kagaku" edited by Nagai K, Kawasaki T. Kinpodo. 2005, pp.3 (Section 1.1, Nakakita S.). |
| Copyrights |
 Attribution-Non-Commercial Share Alike Attribution-Non-Commercial Share Alike
This work is released underCreative Commons licenses
|
| Date of registration:2015-05-08 15:04:45 |
- Yoshizawa, Z., Sato, T., and Schmid, K. (1966) Biochim. Biophys. Acta. 121, 417–420 [PMID : not found]
- Takasaki, S., Mizuochi, T. and Kobata, A. (1982) Hydrazinolysis of asparagine-linked sugar chains to produce free oligosaccharides. Methods in Enzymology 83, 263–268 [PMID : 7098932]
|
This work is licensed under Creative Commons Attribution-Non-Commercial Share Alike. Please include the following citation
How to Cite this Work in an article:
Nakakita, Shin-ichi,
Natsuka, Shunji,
(2015). GlycoPOD https://jcggdb.jp/GlycoPOD.
Web.24,4,2024 .
How to Cite this Work in Website:
Nakakita, Shin-ichi,
Natsuka, Shunji,
(2015).
Release of N-glycans by hydrazinolysis.
Retrieved 24,4,2024 ,
from https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t10.
html source
Nakakita, Shin-ichi,
Natsuka, Shunji,
(2015).
<b>Release of <em>N</em>-glycans by hydrazinolysis</b>.
Retrieved 4 24,2024 ,
from <a href="https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t10" target="_blank">https://jcggdb.jp/GlycoPOD/protocolShow.action?nodeId=t10</a>.
Including references that appeared in the References tab in your work is
much appreciated.
For those who wish to reuse the figures/tables, please contact JCGGDB
management office (jcggdb-ml@aist.go.jp).
|
|